Research Article
Measurement of Resting and Active Aerobic and Anaerobic Metabolism of the Sand Fish, Scincus mitranus at Selected Temperatures
Department of Zoology, College of Science, King Saud University. P.O.Box-2455, Riyadh 11451, Saudi Arabia
Maximal aerobic metabolism is a good indication of the work capacity of several animals (Bennett, 1972 and Hailey et al., 1987). The active oxygen consumption of reptiles has pronounced thermal dependence below their preferred body temperature (PBT), at which it is known to reach maximal levels (Bennett and Dawson, 1976; Bennett, 1972; Al-Sadoon, 1986a; Al-Sadoon and Spellerberg, 1987 and Gatten et al., 1988). Reptile's aerobic scope is strictly temperature dependent, but iguanid and agamid lizards (with large body size) can maximize this scope at PBT (Bennett, 1972), though smaller reptiles generally have greater aerobic scope than the larger ones (Bennett, 1972 and Bennett and Dawson, 1976). Several other activity factors seem to affect the general metabolism of reptiles (Wilson and Gatten, 1989; Wilson et al., 1990 ; Patnaik, 1994 and Thompson and Stewart, 1997). Moreover, anaerobiosis (glycogenolysis or glucolysis to lactic acid) seems to greatly supplement reptile aerobiosis during short outbursts of forced activity (Bennett and Dawson, 1972; Bennett and Licht, 1972; Al-Sadoon and Spellerberg, 1987 and Wickler and Gleeson, 1993). The equation formula of aerobic and anaerobic scopes done by Bennett and Licht (1972), and several other studies, especially on lizards and turtles (Bennett and Dawson, 1972; Al-Sadoon and Spellerberg, 1987 and Wilson et al., 1990) have evaluated the possible total metabolic scope for several species of reptiles.
In present study, the anaerobic contribution to the possible upper limits of activity for the sand fish, S. mitranus was evaluated. Correlation of the thermal dependence with this process was determined and compared with aerobiosis.
Sand fish (skinks) were collected during the summer of 1999 from the sand dunes at north of Riyadh, the capital of Saudi Arabia. All lizards were kept in large tanks with sandy substrata simulating their natural habitat. Food and water were provided ad libitum. The animals were divided into two groups, one of 30 lizards for the determination of aerobiosis, and the other (group of 10) for evaluating the contribution of anaerobiosis during an outburst of forced activity. The animals were fasted for 5-6 days prior to experimentation.
Both processes of active metabolism (aerobic and anaerobic) were calculated and compared by converting their values to adenosine triphosphate (ATP) (Bennett and Licht, 1972) according to the following equations:
1.0 mg lactate formed = 0.0167 mmoles ATP.
1.0 cc oxygen (STP) consumed = 0.2900 mmoles ATP.
Determination of resting and active oxygen consumption: The oxygen consumption rates for resting and active sinks were measured at 15, 20, 25, 30 and 35°C using a double-chamber volumetric system, described by Al-Sadoon and Spellerberg (1985). Active metabolism estimates were obtained from lizards subjected to electrical stimulation by two stainless steel pins used as electrodes and attached to an electric stimulator. These electrodes were implanted subdermally into the abdomen and anterior to the hind limbs inside the volumetric system and shocks (10-14V) were delivered for at least 5 min. Oxygen consumption was determined for every two min during this period and after the animal had been rested for 5 min. The data were expressed as oxygen ml/g body weight/h and corrected to standard temperature and pressure (STP).
Determination of total lactate production in resting and active states: Total lactate concentration was measured using whole body homogenates of resting and active skinks according to the methods of Bennett and Licht (1972) and of Gatten (1985). The data were expressed as mg lactate/g body weight.
Statistical analyses of differences between mean values of various groups of skinks at the various experimental temperatures were carried out by a one-way analysis of variance (ANOVA).
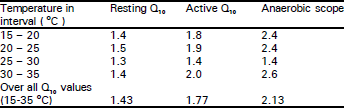
Resting and active oxygen consumption rates: Mean oxygen consumption rate (ml O2/g/h) increased with rising temperatures from 15 to 30 °C, but remained almost stationary at 30-35°C, (Table 1). During the first 1-3 min of stimulation the skinks became hyperactive and their mean rate of O2 consumption (the active O2 consumption phase - VO2) had increased as temperature rose from 15 to 35°C. Aerobic scope increased with rising temperature from 0.184 (ml O2/g/h at 15°C to 0.429 (ml O2/g/h) at 35 °C (Table 1). The highest values of thermal dependence (Q10) at 25-30°C, were 3.3 and 2.3 for resting and active lizards, respectively. The lowest Q10 values were 1.0 and 1.2, respectively at 30-35°C (Table 2).
Resting and active lactate production: The mean values for whole-body lactate expressed as mg/g body weight (resting and active) over the temperature range of 15-35°C, are presented in Table 3.
| Table 1: | Mean oxygen consumption rates ( |
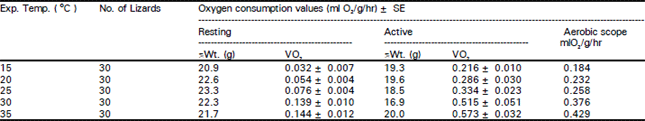 | |
| Table 2: | Thermal dependence (Q10) for resting and active oxygen consumption (VO2) and the aerobic scope of Scincus mitranus at various temperatures |
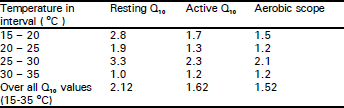 | |
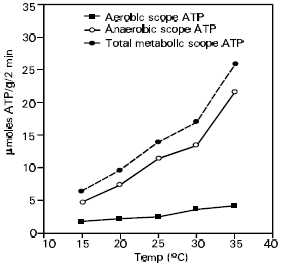 | |
| Fig. 1: | Generation of ATP through aerobic, anaerobic and total metabolic scope during 2- min bursts of activity in the desert lizard S. mitranus. |
There were non-significant (P> 0.05) differences between means of resting lizard’s lactate production over that temperature range (b). Conversely, active lizards had means that increased significantly (P< 0.05) from 0.65 at 15°C to 2.04 at 35°C (a). Anaerobic scope increased with rising temperature from 0.28 mg/g at 15°C to 1.29 mg/g at 35 °C (Table 3). The highest anaerobic scope (84 %) and total metabolic scope were observed at 35°C (Table 4). Non-significant (P> 0.05) differences were observed in the overall Q10 in resting and active lizard’s lactate production (Table 5).
Total metabolic scope during outburst forced activity: Both aerobiosis (1.77-4.14 Fmol/g/2min) and anaerobiosis (4.74-21.54 Fmol/g/2min) increased with rising temperature (15-35°C). Moreover, the total metabolic scope (aerobiosis + anaerobiosis) also increased with the temperature over the same range. The percentage contribution of anaerobic scope (73-84%, at 15-35°C) to the total metabolic scope increased with rising temperature (Fig. 1). Conversely, the percentage contribution of aerobic scope to total metabolic scope decreased with rising temperature (27-16%, at 15-35°C) (Table 4).
Resting and active oxygen consumption of S. mitranus is temperature-dependent. Previous studies on lizards of comparable weights had similar values (Andrews and Pough, 1985; and Al-Sadoon, 1986b). The PBT of the sand fish S. mitranus was found to be 34 -35°C (Al-Johany et al., 1999); and a greater thermal dependence of oxygen consumption had been noticed at low temperature range 15-20 °C (Q10 was 2.8) and the medium temperature range 25-30°C (Q10 was 3.3). Comparable results were obtained from works done on Hemidactylus turcicus , Ptyodactylus hasselquistii (Zari, 1992) and Chamaeleo calyptratus (Zari, 1993).
An equation relating weight-specific metabolic rate of 14 reptile species to body size was formulated by Bennett and Dawson (1976). Their predictions of active metabolic rate, with a value of 0.77 mlO2/g/h for lizards of 17 g body weight were similar to our values for skinks of the same body weight at 30°C (0.52 ml/O2/g/h). At 35 °C our lizards (20g body weight) had a different value (0.57 O2 ml/g/h). Comparisons show the observed values of S. mitranus at 30°C and 35°C agree with those predicted by the weight-specific active metabolism equations of Bennett (1982).
Reptiles have a 5:6 ratio of active metabolism to the resting one at the PBT Bennett and Dawson (1976). The skink S. mitranus, whose PBT was 34°C gave a 4-fold factorial scope (ratio of active to resting metabolism) at 35°C. Other species like Dipsosaurus dorsalis had 17-fold ratio (Bennett and Dawson, 1972) and Pseudemys scripta and Terrapene ornata had 20-24-fold ratio (Gatten, 1974). Chalcides ocellatus (Al-Sadoon and Spellerberg, 1987) gave a ratio of 8:5 at its PBT. It is of interest to note that the factorial scope is temperature-independent; at 15°C in our study S. mitranus was able to increase the ratio of active to resting metabolism up to 7-fold, in comparison with 4-fold at 35°C. Comparable results had been observed on Varanus by Bennett and Dawson (1972), on C. ocellatus and Lacerta vivipara by Al-Sadoon and Spellerberg (1985), on C. ocellatus by Al-Sadoon (1986a, 1986b) and on other species by Varnet et al. (1988) and Jiang and Claussen (1993). The overall Q10 values were lower for active metabolism than for resting metabolism, as was similarly found by Dawson (1976).
The aerobic scope for activity of S. mitranus increased directly with temperature from 15°C to 35°C. It appears that this lizard maximizes its aerobic scope at PBT range (maximal aerobic scope was 0.429 O2 ml/g/h at 35°C). This is not surprising, since under natural conditions most reptiles are known to be capable of behavioral thermoregulation. At temperatures higher than PBT, the aerobic scope of several lizards was temperature independent (Bennett and Dawson, 1972; Wilson, 1974; Bennett et al., 1975; Bennett and Gleeson, 1976; Al-Sadoon, 1986a, 1986b and Al-Sadoon and Spellerberg, 1985, 1987).
| Table 3: | Mean lactate concentration (mg lact/g body Wt.) in whole body homogenates and the anaerobic scope in resting and active Scincus mitranus at various temperatures |
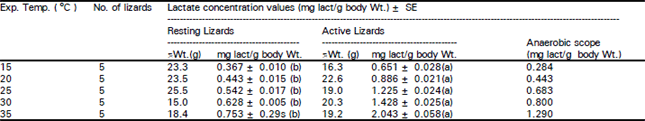 | |
| (a) : Significant (P < 0.05) (b) : Non-significant (P > 0.05) | |
| Table 4: | Aerobic and anaerobic contribution to the total metabolic scope of Scincus mitranus during 2- min bursts of activity |
 | |
| Table 5: | Thermal dependence (Q10) for resting and active lactate production and the anaerobic scope of Scincus mitranus at various temperatures |
 | |
In present study on skinks the aerobic scope was 0.376 at 30°C and 0.429 at 35°C, whilst the Q10 value was 1.2 at the range 30-35°C. Hence a decline in the aerobic scope at PBT was expected.
An interesting comparison can be made between the activity metabolisms of S. mitranus and C. ocellatus. Both lizards belong to the same family (Scincidae), and live in the same climatic region and their metabolic scopes were measured under similar conditions. The maximal anaerobic scope was less in the case of S. mitranus. Thompson and Withers (1997) working on a similar comparative basis on different species of monitor lizards got comparable differences in their results. S. mitranus has a comparable size to C. ocellatus that inhabits the cultivated areas in Saudi Arabia, but has a high aerobic scope (1.24 O2 ml/g/h) as compared with lower one (0.42 O2 ml/g/h) of S. mitranus at 34°C. This difference can be attributed to the enhanced predation effect, activity, wariness and ability to flee to safety of the latter (Al-Sadoon, 1986b). Defense behaviour proved to be dependent on the body temperature, as had been reported by Hailey and Theophilidis (1987) and Wilson et al. (1990).
The aerobic scope of S. mitranus at all experimental temperatures were lower than those of C. ocellatus and this might be due to the behaviour of these lizards. The sand fish lizard, S. mitranus inhabits sand dunes and achieves its PBT (34°C) by heliothermy and thigmothermy. It escapes from predators by diving in the sand. Hence, it does not require a high aerobic scope, whilst C. ocellatus that lives in the open cultivated areas, not the sandy ones, is at risk of predation. The present work on aerobic metabolic scope is in line with the observations on several other species by Bennett (1972); Bennett and Dawson (1976); and Zari (1993). The works of Al-Sadoon (2001) on Scincus hemprichii that belongs to the same family (Scincidae) gave comparable results to those done on S. mitranus.
Anaerobic activity releases blood lactate production, which leads to its removal by the liver. Increase in blood lactate to its maximum levels might not be reached until minutes or hours after cessation of the diving and activity process in reptiles (Moberly, 1968; Wilson, 1974; Bennett and Licht, 1972). The levels in the present study (Table 3) coincided with those predicted by Bennett and Licht (1972).
Contrary to the aerobic scope, the anaerobic scope and capacity were less temperature-dependent (Table 4). Similar results were also shown by Al-Sadoon (1986b) and Al-Sadoon and Spellerberg (1987) for C. ocellatus.
Anaerobic ATP exceeded the aerobic ATP at all temperatures (Fig. 1) as also found by Bennett (1982), who indicated a sfactor of 2-5 fold. The data in Table 4 are similar to those of Bennett (1982), who gave the range of anaerobic metabolism as 50-75% of the total ATP produced during intense activity. Our present data also agree with the previous observations of Al-Sadoon (1986a, 1986b), Al-Sadoon and Spellerberg (1987) and Wickler and Gleeson (1993).
Larger animals may be better in defending themselves against predators, a situation that justifies their difference in aerobic scope. Anaerobiosis may be independent of body weight (Coulson, 1979; 1987).
The total metabolic scope (ATP) of S. mitranus was greater than that of C. ocellatus. If all the increments could be attributed to muscular activity, then the expectation would be for a differential capacity for performance by the two species, depending on their body temperatures. C. ocellatus (that had a higher metabolic scope) outperformed S. mitranus at all experimental temperatures. Such comparisons may help in the clarification of the present situation, but further work on the thermal dimensions of niche adaptation of various reptile species could verify and open broader spectra for deeper research.
I would like to thank Mr. Nasreddin M. Abdo and Mr. Mohammed Y. Abdul-Rawoof for their technical assistance in the laboratory work and compiling the manuscript.