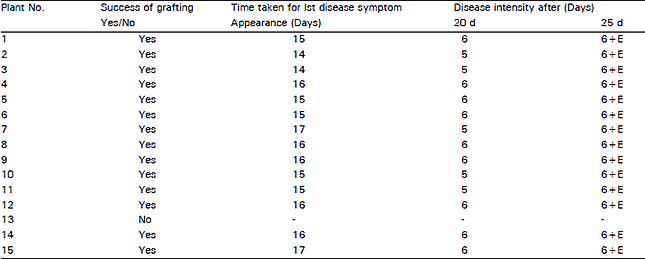
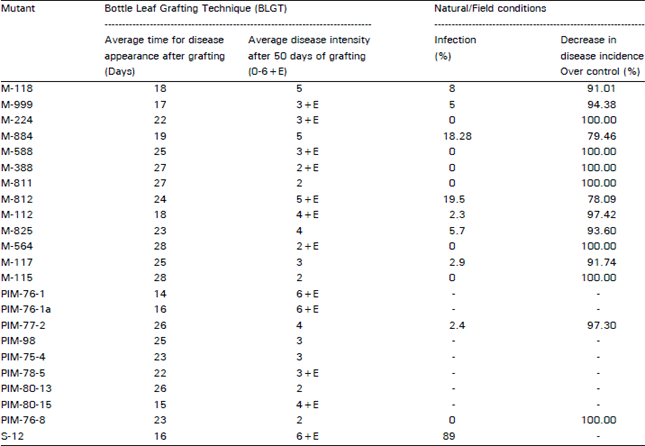
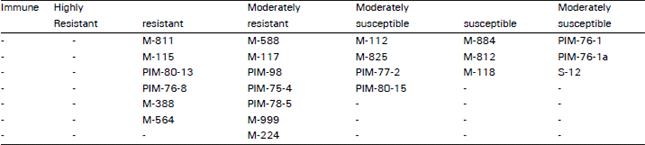
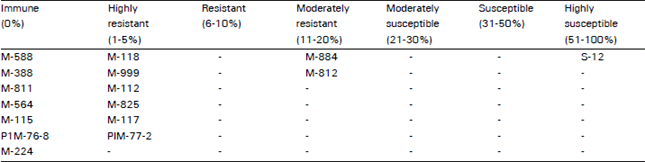
Research Article
Screening of Cotton Mutants for the Resistance Against Cotton Leaf Curl Virus (CLCuV)
Nuclear Institute for Agriculture and Biology (NIAB), Faisalabad, Pakistan
M. Hussain
Nuclear Institute for Agriculture and Biology (NIAB), Faisalabad, Pakistan
A.I. Khan
Nuclear Institute for Agriculture and Biology (NIAB), Faisalabad, Pakistan
M.S.I. Khan
Nuclear Institute for Agriculture and Biology (NIAB), Faisalabad, Pakistan