ABSTRACT
Soybean (Glycine max L.) Cell suspension cultures were exposed to excess copper, in liquid MS-medium. Viability, growth rate and phytochelatins concentration were monitored in treated and untreated cultures. The viability of Cu-treated cultures as reduced and more affected with the time of Cu-exposure, while the O2-uptake was slightly raised in treated cultures and decreased with the time of Cu-exposure. The growth rate was more affected at 10 and 15 days of treatment and reduced by about 76% and 83% below that of the control culture, respectively. The production of PCs in treated cultures markedly increased with the time of Cu-exposure. The glutathione depletion is coincident with PCs production. A marked increase in the level of GSSG in treated and untreated cultures with the time of Cu-exposure. The accumulation of Cu in treated cultures was raised as the time of exposure increased. The accumulation of copper is coincident with PCs levels. Phytochelatins detection seem to be an important biomarker technique in systems contamined with heavy metals.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2000.799.803
URL: https://scialert.net/abstract/?doi=pjbs.2000.799.803
INTRODUCTION
Contamination of soils with ehavy metals becomes more and more a problem in many countries all over the world. In areas where metal contamined soils are used for food crop production, metals relatively mobile within the plant, can easily come into the food chain with great risks for human health. The bioavaliability of heavy metals in soils varies with soils and plant characteristics, e.g., mineralogical and organic matter properties and pH (De Haan et al., 1990), though the metals can also interact mutually. A clear example is the strong Zn-Cd interaction in soils with increasing Zn levels, the availability of Cd in the soil and its uptake by plants are generally inhibited and vice versa (Abdel-Sabour et al. 1988; El-Enany, 1995).
Copper is required by biological systems as a structural and catalytic component of proteins and enzymes and as cofactor (Clijsters and van Assche, 1985). However, excessive ocncentrations of copper are highly toxic (Woolhouse, 1983; Verkleij and Schat, 1990). Copper in the soil operates as a stress facto rin that it caused physiological constraints that decrease the vigor of the plants and even totally inhibit plant growth (Ouzounidou et al., 1994). Glutathione (GSH) identified as a precursor fo rthe ofmration of PCs in plants (Scheller et al., 1987; Steinkamp and Rennenberg, 1985; Schaefer et al., 1997). Treatment of plant cells with buithionine sulfoximine, an inhibitor of γ-Glu-Cys synthetase, reduces the synthesis of PCs and increases sensitivity to metal (Steffens et al., 1986; Reese and Wagner, 1987; Scheller et al., 1987; Salt et al., 1989; Gupta et al., 1998; Gussarsson et al., 1996).
Plants grown under heavy metals stress had increased the levels of sulfhydryl-rich peptides (Phytochelatins), non-protein thiols with the general structure (γ-Gul-cys)n Gly, n varying between 2 and 11 (Grill et al., 1987; Scheller et al., 1987; Steffens, 1990; De Knecht, 1994; Chen et al., 1997). Phytochelatin induction is observed only in plants, algae, yeast and fungi grown under conditions of excess heav metals and have been described as sequester and detoxifiers of heavy metal ions (Zenk, 1996; Skowronski et al., 1998). The phytochelatin compounds were able to detoxify metals inside the cytoplasm of plants cells by in situ chelation (Becher and Hofner, 1994; Stroinski and Zielezinska, 1997; Gupta et al., 1998; Maserti et al., 1998). Phytochelatins also, served as metal carriers during tonoplast transport (Voegel-Lange and Wagner, 1990). Consequently, there is a direct relationship between the PCs levels in plants and elevated metal tolerance (Salt et al., 1989; Min et al., 1997).
The objective of the present work was to study the effect of excess copper on the viability, respiration, growth rate and the usefulness of phytochelatins as a biomarker for copper toxicity in soybean callus cultures.
MATERIALS AND METHODS
Soybean (Glycine max L. Clark) suspension cell culturewere cultured in aseptic conditions in 250 mL conical flasks, containing 50 ml of Ms-medium, supplemented with 0.5 mg/L naphthalene acetic acid (NAA), 105 mg/L benzyladenine (BA), 170 mg/L NaH2PO4. H2O, 100 mg/L inositol, 0.1 mg/L thianmine-HCl, 0.5 mg/L nicotinic acid, 30 g/L sucrose. Half the MS-medium received no additional Cu (control), where the total concentraiton in the other cultures was adjusted to 5 ppm Cu (as Cu SO4). Treatments were given in three replicates. The pH was adjusted to 5.5±0.2 before autoclaving. Cultures were kept on a rotatory shaker at 1000 rpm in an illuminated incubator at temperature 27°C with a light intensity of 376.8 μmol m–2 S–1 provided by fluorescent tube a 16-day/8- night hour photoperiod.
The cells were collected at 2, 5, 15 and 21 days after culture inoculation by vacuum and weighted to determine the fresh weight of each culture.
Viability of cultures: About 200 mg of cells were incubated in five fraction (20-30 mg) in MS-medium containing Cu with 0.8% TTC as 2:1 mixture in 50 mM Na-phosphate buffer (pH =7.4). After 20 hours, the cells were washed with distilled water, the red color was extracted for 1 h at 70°C with 95% heated ethanol. After cooling down, the solution was filtered and measured at 485 nm against 95% methanol as a reference (Towill and Mazur, 1975).
Respiration rata: Respiration rate were measured as uMO2/g fresh weight/min. using a Clark electrode (Type E 1 6WTW, GmbH, Weilheim) in a 3 nil containing temperature constant glass acrylic cuvette, in which the suspension was agitated by a magnetic stirrer.
Extraction and assays of non-protein thiols: Non-protein thiols were extracted as De Vos et al.(1992) by grinding lg callus fresh weight in 1 ml 5% (W/V) sulfosalicylic acid plus 6.3 mM EDTA at 4° C in a mortar. The homogenate was centrifuged at 10.8 xg for 15 min at 4°C. The clear supernatants were collected and immediately used for the determination of thiols. The level of total-SH was determined with Ellman's reagent (Ellman, 1959). One-hundred microliters of supernatant was mixed with 0.7 mL of 0.5 M K2PO4 (pH =7.5) and 0.1 ml of 10 mM 5,5-dithiobis(2- nitrobenzoic) acid. The absorbency was measured spectrophotornetrically after 5 min., at wavelength 412 nm against a blank with 100 μl H20. GSH and GSSG were assayed by the GSSG-recycling method (Anderson, 1985). The final concentration of GSH reductase was 0.5 U/ml in the assay medium of total glutathione (GSH+GSSG) and 1 U/ml in the assay of GSSG. GSSG was used as a standard. Phytochelatin concentrations in supernatant were determined indirectly by substracting, the amount of total glutathione (GSH+GSSG) from the amount of total-SH. Phytochelatins content was expressed as PC-SH in μmol/g f.wt. (De Vos et al.,1992).
Metal measurements: Copper content was determined by atomic absorption spectrop-hotometry, after digest the cells of soybean suspension culture by the method adapted by Vymazal (1984) using acid mixture (36% HCI:20% HCIO4: H2O =2:1:1 vlv).
RESULTS AND DISCUSSION
The viability of soybean cultures treated with toxic level of Cu (5 ppm) and untreated culture (control) was shown in (Fig. 1a). Viability was more affected with the time of Cu-exposure. After 57 days of treatment, the viability of cultures was reduced after 2 and 5 days by about 21.0 and 63% of control, respectively. It was found that the viability depend on the concentration of the metal ion in the culture medium as well as on the period of exposure (El-Enany, 1992). Domazlicka and Opatrny (1989) found that 100 μM Cd resulted in a steep decrease in cell viability of tobacco cells. Jackson et al. (1984) reported that metals inhibited cell division and DNA biosynthesis.
The rate of 02-uptake (dark respiration) was slightly raised in treated cultures (Fig. 1b). A severe inhibition of 02-uptake in treated culture was observed at 15 and 21 days of growth in treated and untreated culture. These results are in agreement with many investigators. The fresh weight of soybean cell suspension culture was shown in (Fig. 1c). The data revealed that Cu reduced the growth rate of the soybean cell suspension, growth was more affected at 2 and 5 days and reduced by about 76.9% and 83% below that of control, respectively. While the growth rate after 10 and 15 days were reduced by about 59, 52% and 55% of control, respectively.
Singh et al. (1989) reported that the addition of Ni, Hg and Cu inhibited the 02 uptake in the cells of Cylindrospermum. In this respect Issa et al. (1995), reported that the response of respiration was quite similar to that of photosynthesis and was enhanced proportionally to metal toxicity on growth, i.e., enhanced respiration with retarded growth. Bittel -et al. (1974) and Lee et al. (1976) concluded that heavy metals inhibit phosphorylation mechanisms and interfered with SH groups of respiratory enzymes.
The effect of the toxic level of Cu (5 ppm) on the levels of acid soluble SH compounds (total SH) was studied in a 21-day experiment (Fig. 2a). Cupper treatment resulted in a marked increase of total-SH level as the time of Cu-exposure increased. The level of total SH increased by about 58% of control during the time of Cu- treatment. While, in control cultures the levels of total-SH varied during the subsequent of growth.
In contrast reduced glutathione (Fig. 2b) show a marked depletion with the time of Cu-exposure. Although the GSH was increased in control culture especially after 5 days of treatment. The GSH depletion is coincident with the increase in PC-SH synthesis (Fig. 3a) especially at 15-days of Cu-treatment. PCs synthesis was lowered at 21-day of Cu-treated cultures. In control cultures the level of PCs was lowered at 15 and 21 day of growth. The oxidized glutathione (GSSG) level was also followed (Fig. 4b).
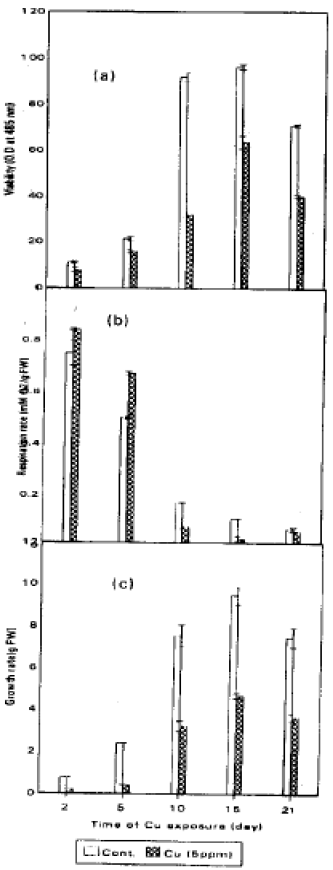 | |
| Fig. 1: | Viability, respiration and growth rate of soybean suspension cultures exposured to 0 and 5 ppm Cu for 2, 5, 15 and 21 days. Values are means with SD (n = 3) |
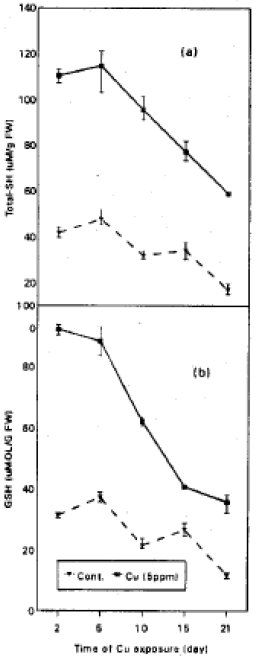 | |
| Fig. 2: | Total -SH and GSH in soybean suspension cultures exposed to 0 and 5 ppm Cu for 2, 5, 10, 15 and 21 days. Values are means with SD = in = 3) |
GSSG contents of suspension cultures of soybean were increased in treated and untreated cultures, The accumulation of GSSG was obviously raised at 21-day of growth.
Glutathione is the most important non-protein thiol present in plants. It is obvious that GSH plays a central rolole in the removal of toxins generated during cellular metabolism (Halliwell, 1978). In plant cells the glutathione is present predominantly in reduced form and its function are connected with thiol group, reactivity of which has important reductive properities. It is generally assumed that GSH maintain cysteine, homocysteine and proteins in biologically active form.
Our results indicate that GSH depletion is coincident with the increase in PCs levels. This is in agreement with many investigators (Scheller et al., 1987; Grill et al., 1988; Mendum et al., 1990). GSH has also been identified as a precursor for the formation of PCs in plants.
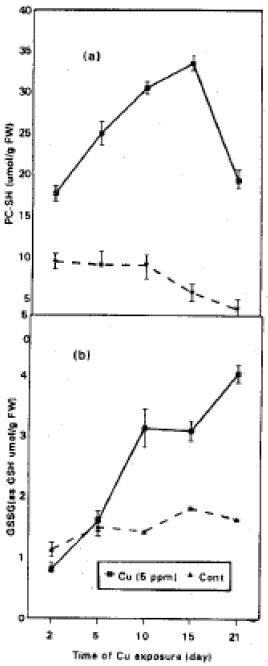 | |
| Fig. 3: | Phytochelatins (a) and GSSG (b), in soybean suspension cultures exposed to 0 and 5 ppm Cu for 2, 5, 10, 15 and 21 days. Values are means with SD =(n =3) |
Treatment of plant cells with buthionine sulfoximine, an inhibitor of y-GluCys synthetase, reduces the synthesis of PCs and increases sensitivity to Cd (Steffens et al., 1986; Reese and Wagner, 1987; Scheller et al., 1987). Chen et al.(1997) found that PC synthase, isolated from tomato cell culture, requires GSH or PCs as a substrates but can not utilize y -GluCys or GSSG . They also 'Concluded that PCs synthase is activated in vitro and in vivo by heavy metals.
In a number of previous studies of PC-SH levels has been assessed by subtracting the GSH concentration from the total acid soluble concentrations (Scheller et al., 1987; De Vos et al., 1992). Our results indicate that PCs production increased with the time of Cu-exposure. This result suggest that copper tolerance in the cells is dependent on an elevated production of PCs. In this respect (Grill et al., 1987; Salt et al., 1989; Mendum et al.,1990) observed that BSO increased the sensitivity of tolerant plant to copper. They also concluded that PCs play a crucical role in copper tolerance.
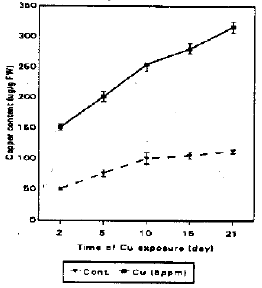 | |
| Fig. 4a: | Cupper accurnulatiion in soybean callus cultures exposed to (0 and 5 ppm Cu) for 21-days. Values are means with SD (n = 3) |
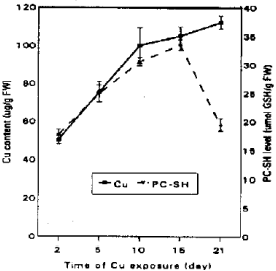 | |
| Fig. 4b: | The relatiionship between PC-SH levels and copper accumulation in soybean callus cultures exposed to (5 ppm) Cu for 21-days. Values are means with SD (n=3) |
Copper in PCs is bound as Cu-thiolate complex depend on the chain length of PC-SH (Reese et al., 1988; Mehra and Winge, 1988). The decline of PC-SH in the cell soybean cultures after 15-day of Cu-treatment may be due to oxidation of. GSH. Phytochelatins released Cu ions , which may result in an increased toxicity. Chen et al., (1997) found that the level of PCs synthase activity was regulated during the cell culture cycle, with the highest activity occurring 3-days after subculture.
Our results in Fig. 3b show a marked increase in the level of oxidized glutathione (GSSG) with the time of culture growth in treated and untreated cultures. The GSH/GSSG status is sensitive to the Redox State of a cell. The increase of GSSG content of cells indicates an increased of oxidation of GSH in vivo. Copper is known to catalyze not only the oxidation of GSH, but also that of other cellular thiols (Miller et al., 1990; Salhany et al., 1978). De Vos et al. (1992) found that, copper capable of catalyzing the oxidation of PC-SH group, as well, which explain the depletion in the level of PCs with the time of Cu-exposure. Stroinski and Zielezinska (1997) found that oxidative stress produced by Cd-stress in potato tuber, activated GSH synthesis and thus suggest a possibility of redox type regulation.
Figure 4a show the accumulation of Cu in treated cultures. Cupper was raised in the cells as the time of growth increased. There is a relation between the internal PC-SH and metal accumulation. This may be the result of a continuous Cu accumulation in storage compartment e.g. (vacuoles) as postulated by Rauser and Ackerley (1987), Vasquez et al. (1992), Voegeli-Lange and Wagner (1990). The actual metal concentration in the cytosol rather than the total metal concentration will determine the actual PC-SH level in plant tissue (Keltjens and van Beusichem, 1998).
Organisms grow on environment polluted with heavy metal are often not stressed to the extent of severe growth reduction. PCs are induced in detectable levels, therefore of PCs consider as a good biomarker technique for systems contamined with heavy metals.
REFERENCES
- Abdel-Sabour, M.F., J.J. Mortvedt and J.J. Kelsoe, 1988. Cadmium-zinc interactions in plants and extractable cadmium and zinc fractions in soil. Soil Sci., 145: 424-431.
Direct Link - Anderson, M.E., 1985. Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol., 113: 548-555.
PubMedDirect Link - Becher, M. and W. Hofner, 1994. Nachweis einer Cd‐komplexierenden, phytochelatinverwandten Verbindung in Scenedesmus subspicatus und Zea mays L. J. Plant Nutr. Soil Sci., 157: 87-92.
CrossRefDirect Link - Chen, J., J. Zhou and P.B. Goldsbrough, 1997. Characterization of phytochelatin synthase from tomato. Physiol. Plant., 101: 165-172.
CrossRefDirect Link - Clijsters, H. and F. van Assche, 1985. Inhibition of photosynthesis by heavy metals. Photosynth. Res., 7: 31-40.
CrossRefDirect Link - De Vos, C.H., M.J. Vonk, R. Vooijs and H. Schat, 1992. Glutathione depletion due to copper-induced phytochelatin synthesis causes oxidative stress in Silene cucubalus. Plant Physiol., 98: 853-858.
Direct Link - Domazlicka, E. and Z. Opatrny, 1989. The effect of cadmium on tobacco cell culture and the selection of potentially Cd-resistant cell lines. Biol. Planta., 31: 19-27.
CrossRefDirect Link - El-Enany, A.E., 1995. Alleviation of cadmium toxicity on maize seedlings by calcium. Biol. Planta., 37: 93-99.
CrossRefDirect Link - Ellman, G.L., 1959. Tissue sulfhydryl groups. Arch. Biochem. Biophys., 82: 70-77.
CrossRefPubMedDirect Link - Grill, E., E.L. Winnacker and M.H. Zenk, 1987. Phytochelatins, a class of heavy-metal-binding peptides from plants, are functionally analogous to metallothioneins. Proc. Natl. Acad. Sci. USA., 84: 439-443.
Direct Link - Grill, E., J. Thumann, E.L. Winnacker and M.H. Zenk, 1988. Induction of heavy-metal binding phytochelatins by inoculation of cell cultures in standard media. Plant Cell Rep., 7: 375-378.
PubMedDirect Link - Gupta, M., R.D. Tripathi, U.N. Rai and P. Chandra, 1998. Role of glutathione and phytochelatin in Hydrilla verticillata (I.f.) royle and Valusneria spiraus L. under mercury stress. Chemosphere, 37: 785-800.
CrossRefDirect Link - Gussarsson, M., H. Asp, S. Adalsteinsson and P. Jensen, 1996. Enhancement of cadmium effects on growth and nutrient composition of birch Betula pendula by buthionine sulphoximine (BSO). J. Exp. Bot., 47: 211-215.
Direct Link - Halliwell, B. and C.H. Foyer, 1978. Properties and physiological function of a glutathione reductase purified from spinach leaves by affinity chromatography. Planta, 139: 9-17.
CrossRefDirect Link - Jackson, P.J., E.J. Roth, P.R. McClure and C.M. Naranjo, 1984. Selection, isolation and characterization of cadmium-resistant Datura innoxia suspension cultures. Plant Physiol., 75: 914-918.
Direct Link - Issa, A.A., R. Abdel-Basset and M.S. Adam, 1995. Abolition of heavy metal toxicity on Kirchneriella lunaris (Chlorophyta) by calcium. Ann. Bat., 75: 189-192.
Direct Link - Keltjens, W.G. and M.L. van Beusichem, 1998. Phytochelatins as biomarkers for heavy metal toxicity in maize: Single metal effects of copper and cadmium. J. Plant Nutr., 21: 635-648.
CrossRefDirect Link - Lee, K.C., B.A. Cunningham, G.M. Paulsen, G.H. Liang and R.B. Moore, 1976. Effects of cadmium on respiration rate and activities of several enzymes in soybean seedlings. Physiol. Plant., 36: 4-6.
CrossRefDirect Link - Maserti, B.E., M. Walker, F. Collina and L. Caltavuturo, 1998. Preliminary results on cadmium and mercury tolerance mechanisms in Nicotiana sp. Sci. Total Environ., 213: 273-277.
CrossRefDirect Link - Mendum, M.L., S.C. Gupta and P.B. Goldsbrough, 1990. Effect of glutathione on phytochelatin synthesis in tomato cells. Plant Physiol., 93: 484-488.
PubMedDirect Link - Miller, D.M., G.R. Buettner and S.D. Aust, 1990. Transition metals as catalysts of autoxidation reactions. Free Radical Biol. Med., 8: 95-108.
CrossRefDirect Link - Min, B.K., S.Y. Choi and S.Y. Lee, 1997. Effect of cadmium treatment on the total thiol groups, glutathione and phytochelatin contents in Oenanthe javanica. J. Korean Soc. Applied Biol. Chem., 40: 243-248.
Direct Link - Ouzounidou, G., L. Symeonidis, D. Babalonas and S. Karataglis, 1994. Comparative responses of a copper-tolerant and a copper-sensitive population of Minuartia hirsuta to copper toxicity. J. Plant Physiol., 144: 109-115.
CrossRefDirect Link - Rauser, W.E. and C.A. Ackerley, 1987. Localization of cadmium in granules within differentiating and mature root cells. Can. J. Bot., 65: 643-646.
Direct Link - Reese, R.N. and G.J. Wagner, 1987. Effects of buthionine sulfoximine on Cd-binding peptide levels in suspension-cultured tobacco cells treated with Cd, Zn, or Cu. Plant Physiol., 84: 574-577.
Direct Link - Reese, R.N., R.K. Mehra, E.B. Tarbet and D.R. Winge, 1988. Studies on the γ-glutamyl Cu-binding peptide from Schizosaccharomyces pombe. J. Biol. Chem., 263: 4186-4192.
Direct Link - Salhany, J.M., J.C. Swanson, K.A. Cordes, S.B. Gaines and K.C. Gaines, 1978. Evidence suggesting direct oxidation of human erythrocyte membrane sulfhydryls by copper. Biochem. Biophys. Res. Commun., 82: 1294-1299.
CrossRefDirect Link - Salt, D.E., D.A. Thurman, A.B. Tomsett and A.K. Sewell, 1989. Copper phytochelatins of Mimulus guttatus Proc. Royal Soc. London Ser. B: Biol. Sci., 236: 79-89.
CrossRefDirect Link - Schaefer, H.J., S. Greiner, T. Rausch and A. Haag-Kerwer, 1997. In seedlings of the heavy metal accumulator Brassica juncea Cu2+ differentially affects transcript amounts for γ-glutamylcysteine synthetase (γ-ECS) and metallothionein (MT2). FEBS Lett., 404: 216-220.
CrossRefDirect Link - Scheller, H.V., B. Huang, E. Hatch and P.B. Goldsbrough, 1987. Phytochelatin synthesis and glutathione levels in response to heavy metals in tomato cells. Plant Physiol., 85: 1031-1035.
Direct Link - Singh, D.P., P. Khare and P.S. Bisen, 1989. Effect of Ni2+, Hg2+ and Cu2+ on growth, oxygen evolution and photosynthetic electron transport in Cylindrospermum IU 942. J. Plant Physiol., 134: 406-412.
CrossRefDirect Link - Skowronski, T., J.A. De Knecht, J. Simons and J.A.C. Verkleij, 1998. Phytochelatin synthesis in response to cadmium uptake in Vaucheria (Xanthophyceae). Eur. J. Phycol., 33: 87-91.
CrossRefDirect Link - Steffens, J.C., D.F. Hunt and B.G. Williams, 1986. Accumulation of non-protein metal-binding polypeptides (γ-glutamyl-cysteinyl)n-glycine in selected cadmium-resistant tomato cells. J. Biol. Chem., 261: 13879-13882.
Direct Link - Steffens, J.C., 1990. The heavy metal-binding peptides of plants. Ann. Rev. Plant Biol., 41: 553-575.
CrossRefDirect Link - Steinkamp, R. and H. Rennenberg, 1985. Degradation of glutathione in plant cells: Evidence against the participation of a γ-glutamyltranspeptidase. Zeitschrift fur Naturforschung C, 40: 29-33.
Direct Link - Stroinski, A. and M. Zielezinska, 1997. Cadmium effect on hydrogen peroxide, gluthatione and phytochelatins levels in potato tuber. Acta Physiol. Planta., 19: 127-135.
CrossRefDirect Link - Towill, L.E. and P. Mazur, 1975. Studies on the reduction of 2,3,5-triphenyltetrazolium chloride as a viability assay for plant tissue cultures. Can. J. Bot., 53: 1097-1102.
CrossRefDirect Link - Vasquez, M.D., C. Poschenrieder and J. Barceleo, 1992. Ultrastructural effects and localization of low cadmium concentrations in bean roots. New Phytol., 120: 215-226.
Direct Link - Voegel-Lange, R. and G.J. Wagner, 1990. Subcellular localization of cadmium and cadmium-binding peptides in tobacco leaves: Implication of a transport function for cadmium-binding peptides. Plant Physiol., 92: 1086-1093.
PubMedDirect Link - Vymazal, J., 1984. Short-term uptake of heavy metals by periphyton algae. Hydrobiologia, 119: 171-179.
CrossRefDirect Link - Zenk, M.H., 1996. Heavy metal detoxification in higher plants-a review. Gene, 179: 21-30.
CrossRefPubMedDirect Link








