Research Article
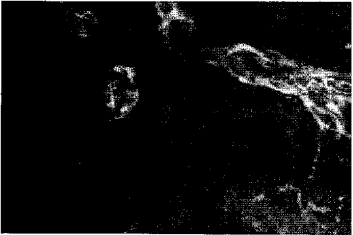
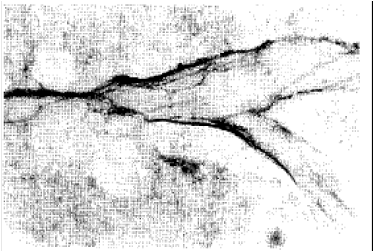
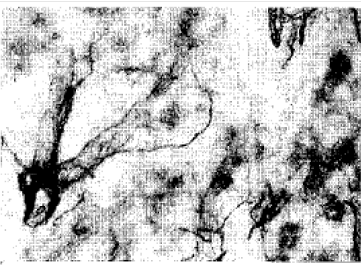
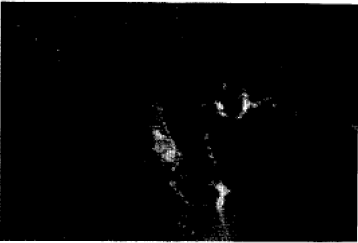
Adrenergic and Cholinergic Innervation of the Pheasant Spleen
Medical Faculty of University P.J. Safarik , Kosice, Slovak Republik, University of Veterinary, Kosice, Slovak Republik
Monika Kocisova
Medical Faculty of University P.J. Safarik , Kosice, Slovak Republik, University of Veterinary, Kosice, Slovak Republik
Maria Sirotakova
Medical Faculty of University P.J. Safarik , Kosice, Slovak Republik, University of Veterinary, Kosice, Slovak Republik
Frantisek Dorko
Medical Faculty of University P.J. Safarik , Kosice, Slovak Republik, University of Veterinary, Kosice, Slovak Republik
Ildiko Skardova
Medical Faculty of University P.J. Safarik , Kosice, Slovak Republik, University of Veterinary, Kosice, Slovak Republik