Research Article
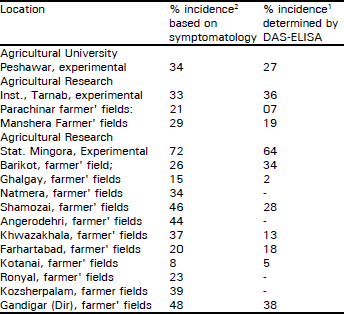
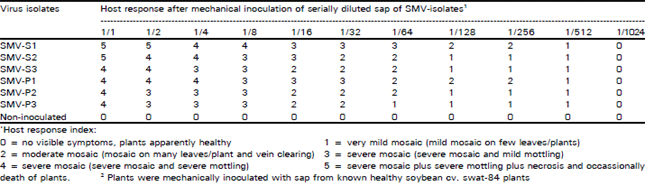
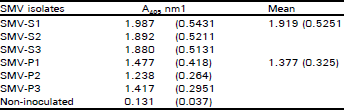
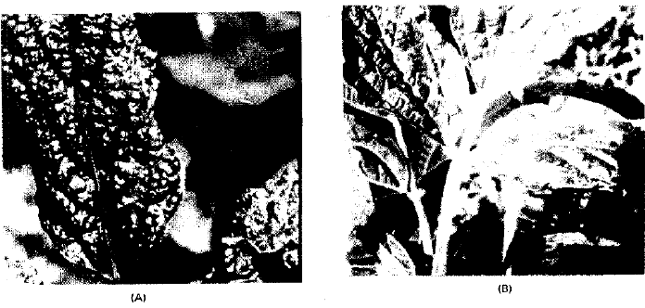
Occurrence and Distribution of Soybean Mosaic Potyvirus in Soybean Crop of North-west Frontier Province, Pakistan and Characterization of Prevalent Isolates
Department of Plant Pathology, Faculty of Crop Protection Sciences, N.W.F.P. Agricultural University, Peshawar, Pakistan
Sher Hassan
Department of Plant Pathology, Faculty of Crop Protection Sciences, N.W.F.P. Agricultural University, Peshawar, Pakistan