Research Article
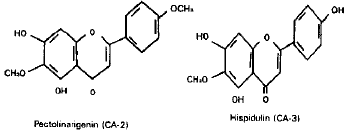
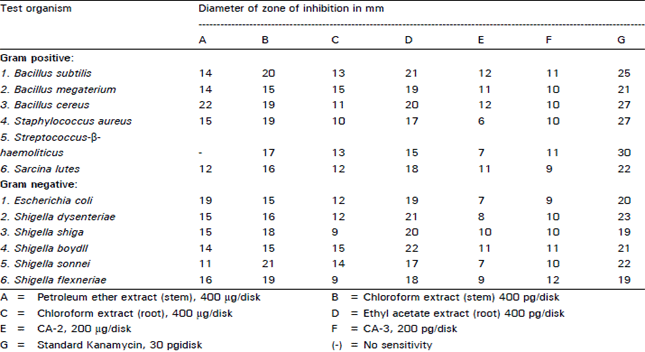
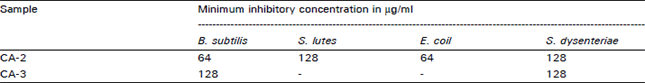
In vitro Antibacterial Principles of Extracts and Two Flavonoids from Clerodendrum indicum Linn
Department of Pharmacy, University of Rajshahi, Rajshahi-8205, Bangladesh
A. T. M. Zafrul Azam
Department of Pharmacy, University of Rajshahi, Rajshahi-8205, Bangladesh
M. A. Gafur
Department of Pharmacy, University of Rajshahi, Rajshahi-8205, Bangladesh