Research Article
Cladosporium spp. An Entomopathogenic Fungus for Controlling Whiteflies and Aphids in Egypt
Economic Entomology Department, Faculty of Agriculture, Mansoura University, Mansoura-35516, Egypt
Whiteflies and aphids are globally important pests because of their direct damage and their role in transmission of plant viruses (Abdel-Baky 1995; Brown, 1998). Beside the appearance of the new whitefly biotypes, silverleaf whitefly (Bemisia argentifolii Bellows and Perring) in the Egyptian entomofauna, which attack different plant hosts with high population levels (Abdel-Baky and Abdel-Salam, 2000; Abdel-Baky et al., 2000). The castor oil whitefly (Trialeurodes ricini Misra) is responsible for the tomato yellow virus in Egypt (Idriss et al., 1997). These lead farmers to apply toxic insecticides frequently.
Chemical control has been confounded by the development of insecticide resistance in both whiteflies and aphids population. The development of high levels of resistance to most chemical insecticides (Dittrich et al., 1990; Kerns and Gaylor, 1992) has forced researchers to look for an alternative means of control (Lacey et al., 1996).
Microbial control agents offer alternatives to chemical pest control and more selective than chemical insecticides. Further, bio-control agents can be integrated with other control methods, they are environmentally more safer than chemically longer and may provide protection after establishment within the host population (Fuxa, 1987).
A number of fungal species have been recognized as promising biocontrol agents for insect pests (Gottel et al., 1990; Ferron et al., 1991). The insect fungi have had many successes because their characteristics (good epizootic, but slow action and over dependence on suitable environmental factors) make them useful after establishment. Many have relatively wide host ranges among insects. Another advantage is the fact that they do not have to be ingested by the insect host but can invade the host upon contact with the cuticle (Boucias et al., 1988).
Among the best fungal pathogens attacking whiteflies known, Paecilomyces fumosoroseus, Beauveria bassiana, Verticillum lecanii and Aschersonia spp. (Lacey et al., 1996). Meanwhile, Pandora neoaphidis, Conidiobolus sp., V. lecanii, B. bassiana, Entomophothora sp. and Neozygotes fresenii are known as aphid entomopathogenic fungi (Hatting et al., 1999). In spite of this, some authors consider Cladosporium spp. as a new promising biological control agent against homopterous insect. De Carvalho et al. (1972) reported that Cladosporium herbarum was associated as a controlling candidate against cashew whitefly, Aleurodicus coccis and also effective as biocontrol agent against three species of whiteflies namely, Bemisia sp., Aleurothrixus sp. and Dialeurodes sp. on various plant hosts in Venezuela (Rojas et al., 1998). Moreover, C. aphids recorded as bio-control agent against Alerurochiton aceris in Finland (Hulden, 1986) and Chionaspis salicis (L.) (Coccoidea: Diaspidididae). In china, C. cladosporioides caused 20-57% natural mortality of Hemiberlesia pitysophila under field conditions and 39% in the laboratory tests (Pan et al., 1989). Whereas, Thumar and Kapadia (1994) mentioned that Cladosporium spp. was able to infect Aleurolobus barodensis nymphs at all year times in India. Han et al. (1997) noticed that the entomopathogenic fungi, Cladosporium spp. naturally occurred and caused epizootic infection to the population of Aleurocanthus spiniferus in China.
Regarding the role of Cladosporium spp. against aphids, there are number of reports emphasize the ability of such fungus in reducing the population size of these pests (Lagowska, 1995; Vallejo et al., 1996; Han et al., 1997). In Egypt, Abdel-Baky et al. (1998) recorded three species of Cladosporium (C. uredinicola, C. cladosporioides and C. chlorocephalum) which infect Bemisia spp., Aphis gossypii and Empoasca sp. They reported that the fungus was occurred naturally at high percentage of incidence (10.0-28.0%) under field conditions. They also reported that C. uredinicola was the most dominant one and more virulent in the laboratory tests. The aim of the present investigation is to study the pathogencity and epizootic of Cladosporium spp. and its seasonal occurrence on certain homopterous insects.
Survey sites, plant hosts and insects: The survey was carried out at three regions of Dakahlia Governorate (Mansoura, Talka and Aga). The pests involved in the survey were Bemisia spp., silverleaf whitefly (SLWF), B. argentifolii, castor oil whitefly (CQWF), T. ricini, cotton aphids, Aphis gossypii (Glov.) legume aphids, A. craccivora Koch. and duranta aphid, A. durantae Theobald. SLWF was surveyed on the three plant hosts (squash, cotton and Mexican fire plant, Euphorbia prunifolia Jacq.) while both Bemisia spp. and castor oil whitefly were surveyed only on castor oil plants, as this host was the only specific host for this pest. Cotton aphid (CA) was surveyed on the cotton, cucumber and eggplant. Meanwhile, the legume aphid (LA) was surveyed on cowpea and duranta aphid (DA) was surveyed on duranta plants.
Sampling Protocols
Whiteflies:
The silverleaf whitefly (SLWF): Twenty-five plants were chosen at random presenting plants of corner and center of the field. The sample was prepared by selecting five leaves from each plant (two leaves from each upper and lower third and one from the middle third of the main stem). Leaves from each group were removed and put in a plastic bag and transferred to the laboratory for examination. Eggs, small nymphs and large nymphs of SLWF were investigated under stereomicroscope and counted as a total/leaf. The infected individuals (cadavers) were also counted. This protocol was applied on cotton, squash and Mexican fire plant.
Castor oil whitefly (COWF): Castor oil plants were normally found on the edge of water canals, roads and around the edge of fields in Egypt. Five plants presenting five locations in Mansoura and Talka regions were chosen while the previous program, which was used with SLWF was applied. Since Bemisia spp. and COWF are more preferable to castor oil plants (Abdel-Baky et al., 2000), therefore, the differentiated between these two species was done on the base of pupal case shape and the methods of eggs laid on this host.
Aphids: Because of the newly formed parts of plants are more favorable to the aphid colonies for feeding and development, three leaves from the upper third of the main stem were chosen randomly, removed and put in a plastic bag and transferred to laboratory for investigation. The experimental samples were 75 leaves representing 25 plants of each crop. The same technique used with whiteflies was applied.
Fungal Isolation and Identification: The following two procedures were used in fungi isolation:
| 1- | Insect cadavers showing natural external growth of fungi were collected and maintained in ptri-dishes contain potato dextrose agar (PDA) media. The inoculated ptri-dishes were kept in an incubator at 27±2 °C and 75±5% R. H. until further growth of the fungi. Spores of pure cultures were inspected under a compound microscope. |
| 2- | Predicted insects to be infected due to their abnormal movement were surface-sterilized in a 1% sodium hydrochlorite solution for 30 second and washed in distilled water. Then the insects were cultivated in ptri dishes (25 insects/dish) on PDA media and kept in an incubator under the same regime of temperature and R.H. |
Identification of isolated fungi was done primarily in Plant Pathology Department, Fac, Agric., Mansoura University. Confermintaion of Fungus identification was based on the external symptoms and the morphology of the fungi and then habit characters were used in consultation with Waterhouse and Bradey (1992) and Humber (1997) and Commonwealth Mycology Institute, Kew, Surrey, England (Ellis, 1971, 1976) to confirm the preliminary identification..
Bioassay: Each fungus associated with the insects tested was used to evaluate its pathogenic role on B. argentifolii, A. gossypii and A. craccivora under laboratory condition. Two hundred individuals from each insect species were chosen after surface-sterilized in a 1% sodium hydrochlorite solution for 30 s and washed in distilled water. Each prtidish contained 25 individuals was considered as one replicate. The insects placed in a dark color blotter moist with the fungal suspension with one concentration (10 × 106 spores/ml.) (Abdel-Baky et al., 1998). A piece of plant leaf was added to each prti-dish after sterilization to be a source of food. Vandenberg (1996) and Abdel-Baky et al. (1998) techniques for preparation of tf.e fungal incula were followed. Data collected daily and continued for 7 days.
Statistical Analysis: Analysis of variance, correlation coefficient and stepwise regression models were used for Data analysis (CoStat Software, 1990). Percentages of Cladosporium infected homopterous insects were calculated by dividing the total number of infected insect species with Cladosporium (summed over all samples) by the total recorded numbers of each species sampled then multiplying by 100.
Entomopathagenic fungi species: Five species of fungal pathogens were isolated as native bio-control agents in Dakahlia Governortae from whiteflies and aphids. The fungal names and their incidence percentages are presented in Fig. 1. Data showed that Cladosporium spp. was the most dominant one, which formed 80.87% of the total isolated species under both filed, and laboratory conditions. Fusarium sp. ranked the 2nd place with 7.6% followed by Verticilium lecanii with 5.7% of the total fungi recorded. Meanwhile, incidence of Trichocithium roseum and Epicocum sp. was lower which listed 3.9 and 2.1% of the total, respectively (Fig. 1). Moreover, C. uredinicola formalized over 90% of Cladosporium spp. that found attacking whiteflies and aphids (Fig. 2).
Incidence of Cladosporium spp. on some homopterous insects under field condition: Cladosporium spp. were found naturally attacking most of homopterous insects that exist in Egyptian entomofauna.
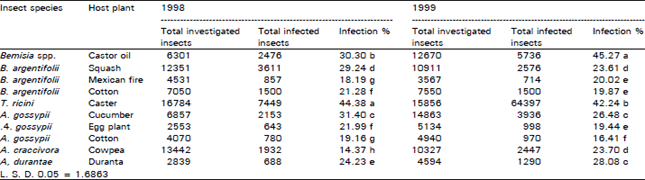
| Table 1: | Percentages of Cladosporiurn spp. incidence on certain homopterous insects at Dakahlia Governorate, Egypt |
 | |
| Means followed by the same letter in a column are not significantly differences (Duncan's Multiple Range Test) | |
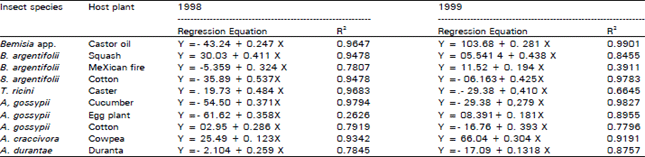
| Table 2: | Stepwise regression analysis between the total investigated insects and total infected by Cladosporium spp. during 1998 and 1999 |
 | |
| Table 3: | Correlation coefficient between the percentages of infected insects by Cladosporium spp. in 1998 and 1999 |
 | |
| Table 4: | Efficacy of five fungal species isolated on three homopterous insects |
 | |
The prevalence varied according to the insect species and the plant host type (Table 1). Regarding its suppression role on whiteflies, the fungus caused high infection rates, which reached 44.38% and 42.24% on castor whiteflies in 1998 and 1999, respectively. The infection percentages of Cladosporium against Bemisia spp. on castor oil plants were high and formed 39.30% in 1998 and 45,27 in 1999. Percentage of SLWF cadavers infected by Cladosporium spp. ranged from 18.19-39.24% in 1998 and 19.87-23.61% in 1999 (Table 1). The lowest rate of infection against SLWF was recorded on both cotton and Mexican fire plants in the two successive years.
A high prevalence of the disease caused by Cladosporium against cotton aphid occurred on cucumber plants (31.40% and 26.48%) in both years, respectively. Meanwhile, the occurrence of the fungus as a natural enemy of A. gossypii on cotton fields was lower 19.16% in 1998 and 16.41% in 1999. The effect of Cladosporium spp. on A. durantae was high 24.23% and 28.08% in both years, respectively, than its effect on A. craccivora (Table 1). The natural occurrence of Cladosporium spp. showed variation on whitefly life stages as it was 87.8, 8.05 and 4.15% for nymphs, adults and eggs, respectively (Fig. 3).
Table 2 shows the regression equations, which express the numerical relationship between the fungus Cladosporium and each of the tested insect pests in the two years of study. The statistical analysis revealed the presence of a high relationship among the fungus and its victims. The results, also, indicate that the fungus has a continuos effect from year to another as seen from Table 3.
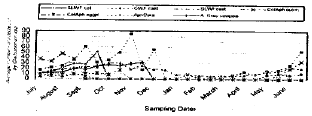
Seasonal colonization of Cladosporium spp. under field condition: Natural occurrence of Cladosporium spp. was observed all year around with different infection rates. Figure 4 and 5 show the seasonal abundance and the yearly distribution of the fungus on the homopterous insects involved in this study. Levels of infection extend from June until the end of November in the two years of study (Fig. 4, 5). The degree of fungus association with both Bemisia spp. and T. ricini on castor oil plants was relatively higher and continued all year around, then decreased to low levels from December until May of each year. The fungus caused an epizootic diseases to whiteflies from Sept. until end of November in both years (Fig. 4, 5). The same trend occurred with the SLWF on cotton plants and the fungus was effective against the pest from July until Sept. in 1998 and 1999. Moreover, Cladosporium incidence on aphid species was high and the infection rates were varied according to aphid species and its population size under field condition.
The fungus prevalence in the field was found associated with the favorable environmental conditions during the period from June until November {temperature average 21.19 (15.73 - 30.43) and R. H. 60.96 (33.80 - 85.67) in both years}.
Laboratory bioassay studies: All fungal species tested were pathogenic to the three insect species tested at conidial concentrations of 10 x 106 spores/ml. Fungal species were significantly differed in virulence to the SLWF, cotton aphid and legume aphid (Table 4). At 3 d, Cladosporium spp. caused the higher mortality in this short time for the insect species than all other fungal species. Sensitivity of SLWF to Cladosporium infection was high as the mortality reached 83.5% than both aphids (A. gossypii 37.5% and A. craccivora 38.0%). Verticillium lecanii was the other second fungus, which caused 37.0, 18.0 and 13.5% mortality in B. argentifolii, A. gossypii and 4. craccivora population, respectively. On the other hand, the efficacy of Tricocithium roseum and Epicocum sp. was low in comparison with the other species tested (Table 4).
The success of the entompathogens in insect control are largely due to epizootiologcal or ecological factors, i.e. the speed of kill and economic injury level (ElL) of the pest, effects of the ecosystem and environment, timing and transmission and host resistance (Fuxa, 1997). Moreover, the efficiency of a natural enemy in bio-control is influenced by the host range, specificity of the fungal pathogen, or strain, the host's physiological state, nutrition, defense mechanisms, cuticle and epicuticular of the pest (McCoy et al., 1988; Boucias et al., 1988). The significance of Cladosporiurn spp. as a one of the effective biological control agents against whiteflies, aphids, and scale insects in the world have been reviewed (De Carvalho et al., 1972; Roberts and Humber, 1981; Hulden, 1986; Pan et al., 1989; Humber, 1991; Thumar and Kopadia, 1994; Han et al., 1997; Abdel-Baky et al., 1998). Farias and Filho (1987), found that Cladosporium spp. was the most important fungus isolated from nymphs of Aleurothrixus aepium in cassava plants. The fungus caused 82.2% mortality of the insect nymphs under field conditions. The percentage Bemisia spp. infected naturally by Cladosporium were varied from 10.0 - 28.0% according to the plant host and over 80% in the laboratory tests (Abdel-Baky et al., 1998). The virulence of Cladosporium varied on the base of insect species and type of plant host (Table 1). Hare and Andereadis (1983) explained the role of plant host on the infection rate of the fungus. They referred to significant differences in susceptibility of Leptinotarsa decemlineata to Beauvera bassiana when reared on four different Solanceae species, considering fungal prevalence in field and mortality in laboratory. This interpretation explains why the infection of the insect was varied within plant species? Whereas, plant morphology may also interfere with insect susceptibility to fungal infection (Hare and Andreadis, 1983; Boucias et al., 1984). The insect species, plant host, the quality of fungal inoculum and the climatic conditions during the survey could account for the differences in percentages of Cladosporium spp. observed between insects.
Data presented in Table 2 might throw some light on the infection relationships, which govern the fungus incidence. This consequence relationship of fungus infection under field condition might be possible to utilize in controlling the insect pests. The relationship between all infected insects in two years of study was very high (R =0.8759 ±0.093) and indicates that the degree of correlation between the two variables showed stronger relationship that is actually present under the field condition (Table 3).
Entomopathogenic fungi in nature cause a regular and tremendous mortality of the pests (Steinhaus, 1949). The seasonal distribution of Cladosporium sop, mostly tends to be high from June to November (Fig. 4, 5) and coincide with population of the insect. Furthermore, the increase in whiteflies or aphids population followed by an increasing in the infection level if climate conditions and ecological factors were favorable for the fungus (Carruthers and Soper, 1987; Abdel-Baky et al., 1998).
The high incidence of Cladosporium infection on the castor oil plants with Bemisia spp. and T. ricini in 1998 and 1999 (Table 1) may be due to one or more of the following reasons: 1) castor oil plants cultivated and grown on the edges of fields or/and on the border of water canals, whereas water evaporation's are higher enough to spore germination and widespread of fungal spores, 2) increase the fogging particularly in the end of the summer and fall and 3) whiteflies immature harbor on the lower surfaces of plant leaves while the nymphs prefer the lower leaves on the main stem of plant. All these ecological factors provide humidity around both insect and fungus, which increase the infection of Cladosporium and cause the epizootic. The results are in agreement with number of reports which shows the role of natural field epizootics followed periods of high rainfall or relative humidity and link disease outbreaks with ambient moisture (Hajek and St. Leger, 1994; Carruthers et al., 1997).
Gottel et al. (1990) pointed out that fungi with wide host ranges are frequently and even more specific under field condition. Such specificity is thought to be due to the complex biotic and abiotic interactions the field. This is in agreement with the results obtained in this study (Table 4)
The perennial agroecosystem offer a permanence in vegetation that could potentially allow host and pathogen populations to persist in some degree balance (Franz, 1971). This could be lead to residual pathogen activity or widespread infection, called an epizootic, because the fungus can multiply and persist on its host insect (Ferron, 1981). The high epizootic potential of Cladosporium spp. which observed on both whiteflies and aphids populations can be attributed to many factors which, when considered in total, describe a pathogen well adapted to parasitism of a highly mobile, rapidly developing host which attains high population densities during favorable periods of spring, summer and fall.
 | |
| Fig. 1: | Percentages of five-entomophatogenic fungi incidence isolated from whiteflies and aphids in Dakahlia Governorate, Egypt |
 | |
| Fig. 2: | Percentage of Cladosporium incidence under field condition |
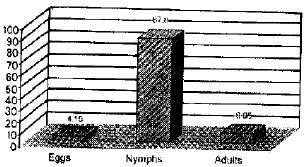 | |
| Fig. 3: | Percentage of Cladosporium spp. infection against whitefly life stages under field condition |
 | |
| Fig. 4: | Seasonal incidence of Cladosporium spp. on six homopterous species in 1998 |
 | |
| Fig. 5: | Seasonal incidence of Cladosporium spp. on six homopterous species in 1999 |
In conclusion, Cladosporium spp. appears to be one of the most effective native bio-control agents against homoptrous insects in Egypt. However, to achieve good control treatments, it must be incorporated into IPM program
Thanks are due to Drs. A. M. Abou El-Naga, M. A. El-Ad!, A, A. Gahinm, Economic Entomology Dept., Fac. Agric., Mansoura Unv. for their sincere guidance and reviewing the manuscript. Also grateful to Dr. A. H. Abdel-Salam for his helping, critical revision and statistical analysis. Also, My special thanks to Dr./ M. A. El-Wakil and Nehal S. Arafat Plant Pathology Dept., for advice, cooperation, using the Lab. facilities and fungal identification.