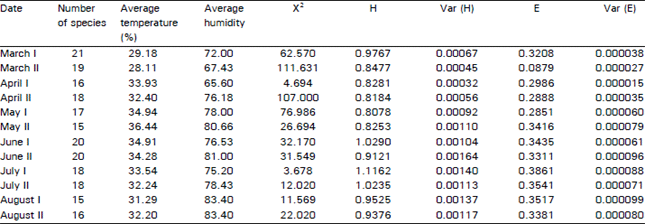
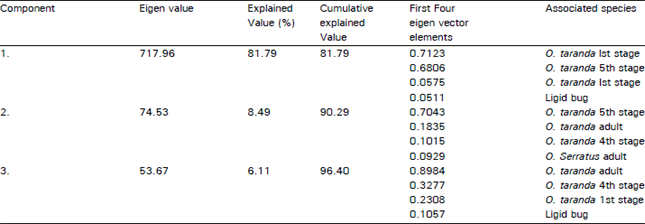
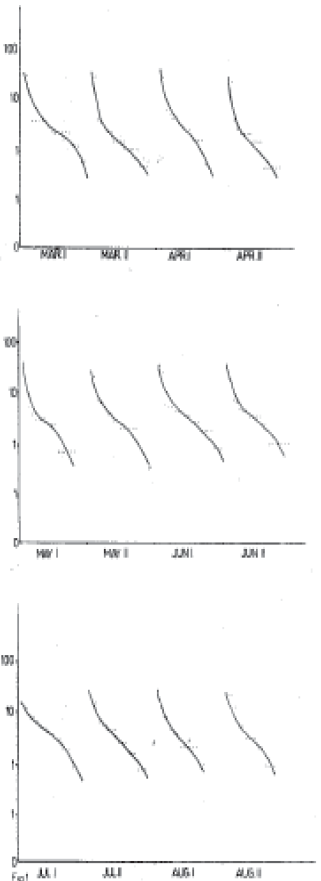
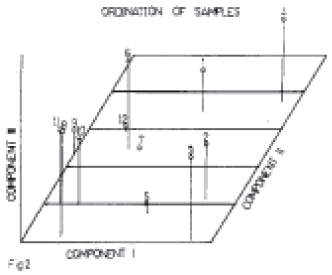
Research Article
Multivariate Analysis of a Tropical Insect Community
Department of Zoology University of Karachi, Karachi-75270, Pakistan
S. Shahid Shaukat
Department of Botany, University of Karachi, Karachi-75270, Pakistan
Shaheen Abidi
Department of Zoology University of Karachi, Karachi-75270, Pakistan