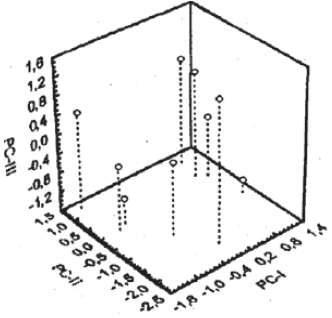
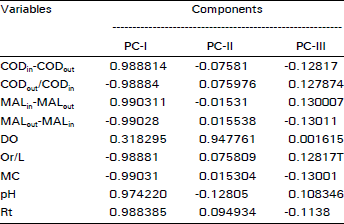
Research Article
An Application of Principal Component Analysis to The Study of Activated Sludge Treatment System Performance Efficiency For the Degradation of Malathion
Department of Botany, University of Karachi, Karachi-75270, Pakistan
S. Shahid Shaukat
Institute of Environmental Studies University of Karachi
Moazzam Ali Khan
Department of Botany, University of Karachi, Karachi-75270, Pakistan
M. Altaf Khan
Department of Botany, University of Karachi, Karachi-75270, Pakistan