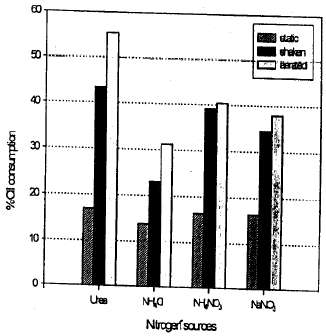
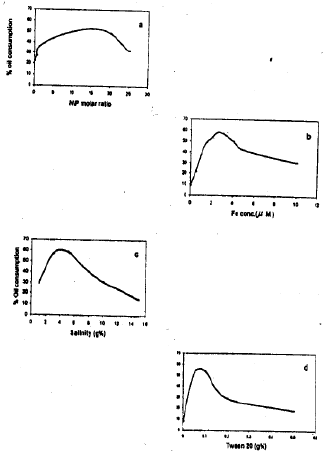
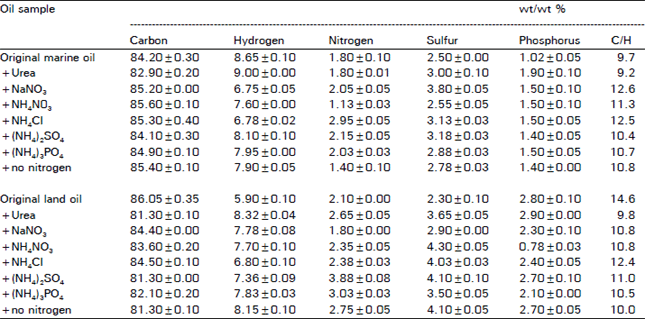
Research Article
Crude Oil Biodegradation by Naturally Inhabiting Mixed Bacterial Culture under Different Environmental Factors
Botany Department Faculty of Science, Cairo University Giza, Egypt
Nagwa M. Morsi
Botany Department Faculty of Science, Cairo University Giza, Egypt
Magdy El-Said Mohamed
Botany Department Faculty of Science, Cairo University Giza, Egypt