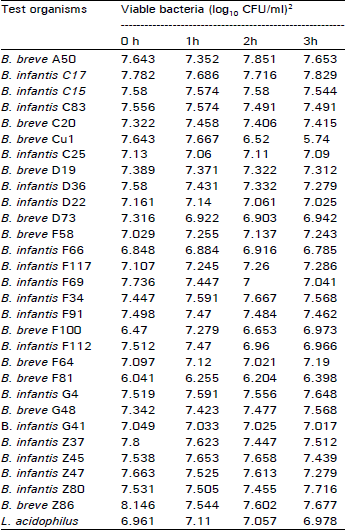
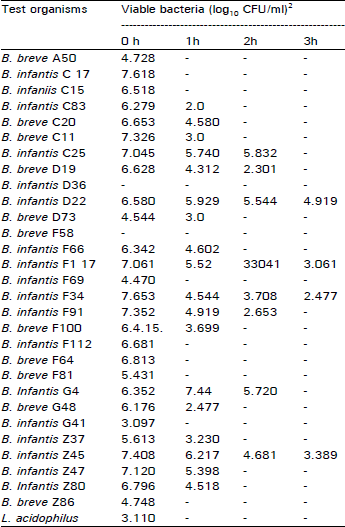
Research Article
Acid Adaptation of Bifidobacteria Isolated from Infant Stools to Simulated pH of Human Stomach
Department of Food Technology, Faculty of Food Science and Biotechnology, Universiti Putra Malaysia, Serdang, Malaysia
A. M. Yazid
Department of Food Technology, Faculty of Food Science and Biotechnology, Universiti Putra Malaysia, Serdang, Malaysia
A.M. Ali
Department of Biotechnology, Faculty of Food Science and Biotechnology, Universiti Putra Malaysia, Serdang, Malaysia
M.H. Ghazali
Department of Biotechnology, Faculty of Food Science and Biotechnology, Universiti Putra Malaysia, Serdang, Malaysia
H. Zaitun
Department of Food Science, Faculty of Food Science and Biotechnology, Universiti Putra Malaysia, Serdang, Malaysia
N.A.Nur Atiqah
Paediatric Institute, Hospital Kuala Lumpur, Jalan Pahang, 50586, Kuala Lumpur, Malaysia