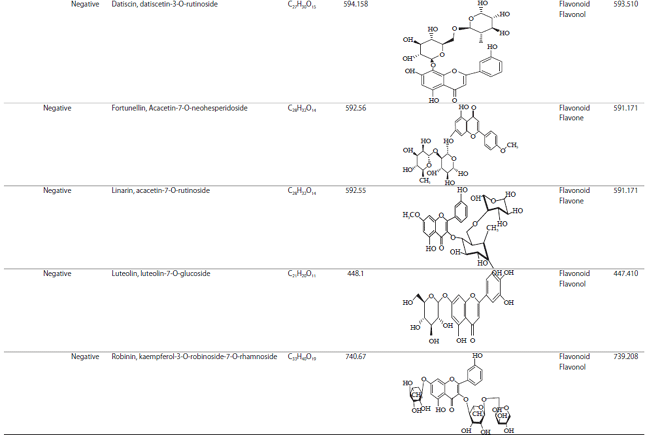
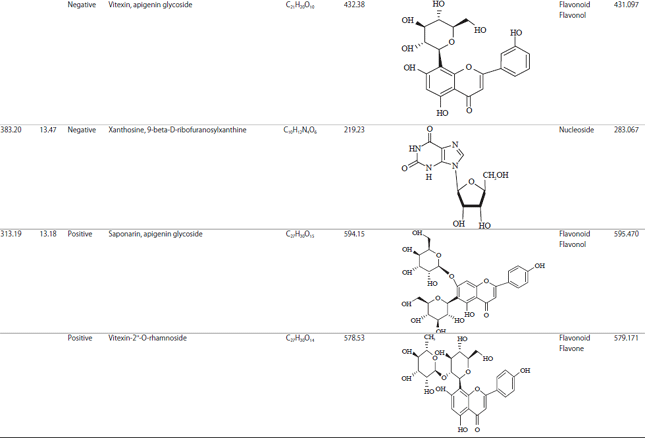
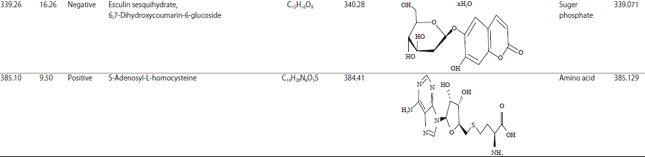
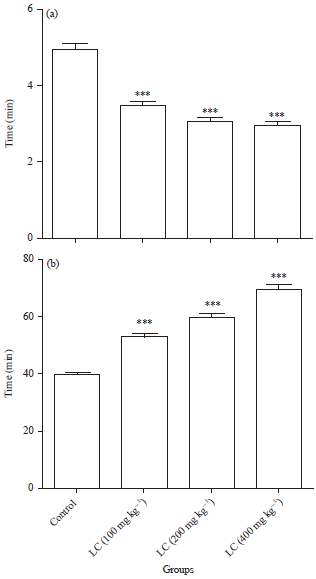
Research Article
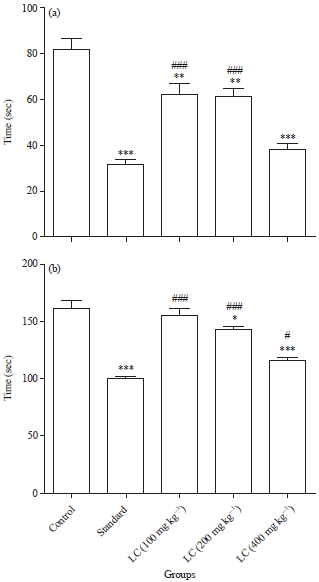
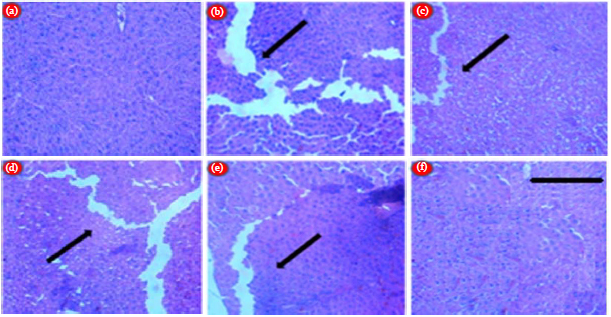
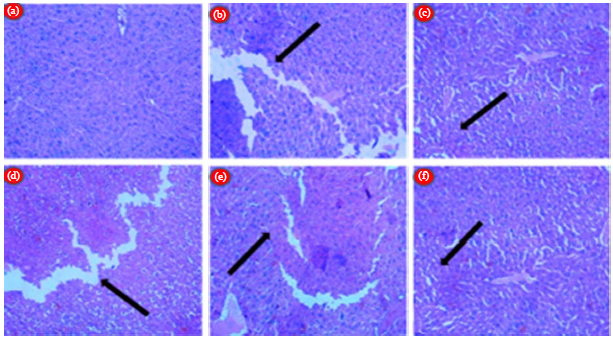
LC-ESI MS/MS Profiling, Antioxidant and Anti Epileptic Activity of Luffa cylindrica (L.) Roem Extract
Department of Pharmaceutical, Engineering and Technology, Indian Institute of Technology, Banaras Hindu University, Varanasi, 221005 Uttar Pradesh, India
LiveDNA: 91.23132
Bhoosan Yadav
Department of Pharmaceutical, Engineering and Technology, Indian Institute of Technology, Banaras Hindu University, Varanasi, 221005 Uttar Pradesh, India
Prabhat Upadhyay
Department of Pharmacology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, 221005 Uttar Pradesh, India
LiveDNA: 91.23098
Pradeep Kumar
Department of Botany, MMV, Banaras Hindu University, Varanasi, 221005 Uttar Pradesh, India
Chandrashekhar Singh
Department of Botany, MMV, Banaras Hindu University, Varanasi, 221005 Uttar Pradesh, India
Jyoti Dixit
Department of Botany, MMV, Banaras Hindu University, Varanasi, 221005 Uttar Pradesh, India
Kavindra Nath Tiwari
Department of Botany, MMV, Banaras Hindu University, Varanasi, 221005 Uttar Pradesh, India
LiveDNA: 91.986