Research Article
Molecular Modelling Analysis of the Metabolism of Cocaine
School of Biomedical Sciences, Faculty of Health Sciences, The University of Sydney, Australia
Cocaine (benzoylmethylecgonine) is one of the main alkaloids of Erythroxylon coca (Clauwaert and Lambert, 1995) that has a long history of human use and abuse (Pavlova et al., 2004). It is an alkaloid ester with a rigid structure found in the leaves (at a concentration of 1%) of the shrub Erythroxylon coca that grows at elevations between 0.7 to 2.4 km in the Andes Mountains (Smith and Reynard, 1992). Whereas cocaine hydrochloride is highly soluble in water, free cocaine base is highly lipid soluble and therefore passes more readily across the mucous membranes, such as those in oral-pharyngeal cavity. Cocaine acts as a local anaesthetic and stimulant causing increased alertness and a sense of euphoria. Being a vasoconstrictor, cocaine is used as an analgesic in ear, nose and throat surgery where it will lessen bleeding from highly vascularised sites. Sustained abuse of cocaine as a recreational drug is widespread around the world (Ladona et al., 2000). Despite a decline in the number of cocaine users between 1985 and 1992, the abuse of the drug in the United States has not decreased further during the past decade (Paula et al., 2004). In 1997, the number of cocaine users in the United States was estimated at 1.5 million. The extreme danger and high mortality rate resulting from cocaine abuse has attracted the attention of physicians and the analysts. Cocaine has been reported to cause toxicity mainly to cardiovascular system and to a lesser extent to the liver (Perino et al., 1978). Because it is known that cocaine can cross placenta (Schenker et al., 1993) the abuse of the drug during pregnancy is of major concern in certain countries where pregnant women take several drugs along with cocaine.
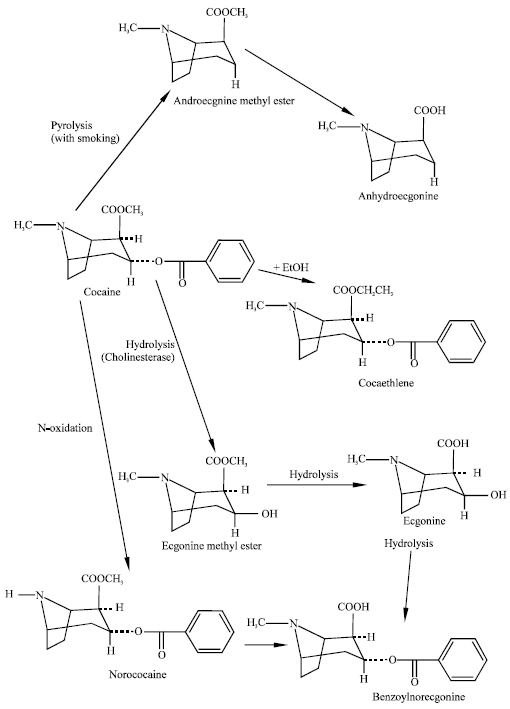 | |
| Fig. 1: | Metabolic pathways for cocaine (Pavlova et al., 2004) |
The pharmacological effects of cocaine are mediated through norepinephrine, dopamine and serotonin neurotransmitter system (Gold et al., 1985). Cocaine produces an accumulation and prolongation of the effects of these transmitters by blocking the presynaptic reuptake of these sympathomimetic transmitters (Rofael and Abdel-Rahman, 2002).
Cocaine is extensively metabolised in humans to pharmacologically inactive metabolites ecgonine methyl ester (ECG), benzoylecgonine (BE) and ecgonine (ECG). Only a small percentage is excreted unaltered in urine (Inaba et al., 1978). Among the metabolites BE has six times longer half-life than cocaine. Norcocaine is a relatively minor metabolite in humans (Schindler et al., 2001). Three enzymes that are involved in cocaine metabolism in humans are: human carboxylesterase 1 (hCE1), intestinal CE (also termed human CE2) and serum butyryl-cholinesterase (hBChE). Human carboxylesterase 1 is a serine hydrolase found in the liver, small intestine, kidney, lung, testes, heart, monocytes, macrophages and circulating plasma (Satoh and Hosokawa, 1998). Whereas CE and hBChE hydrolyse the benzoyl ester linkage in cocaine, only hCE1 hydrolyses the methyl ester linkage to generate benzoylecgonine (Bencharit and Morton, 2003). The enzyme offers a significant promise in the treatment of cocaine overdose as it has the ability to bind to two cocaine molecules at the same time to produce the primary metabolite of cocaine.
Several recent studies reported that alcohol can interfere with cocaine metabolism. When cocaine and alcohol are abused together, hCE1 produces the toxic metabolite cocaethylene from cocaine (Shuster et al., 1977). Pharmacological, physiological and behavioural properties of cocaethylene suggest that it possesses similar characteristics as those of cocaine. CYP3A isoenzymes also play an important role in cocaine metabolism in human liver microsomes and this may be of importance in foetal toxicity associated with maternal abuse of cocaine during pregnancy. Although the Km values of CYP3A enzymes found in adult livers are high, it has been suggested that this pathway would become more significant in circumstances of CYP3A induction and drug interactions leading to liver toxicity in chronic abusers of cocaine. Figure 1 summarizes the major metabolic pathways of cocaine in humans. The pathway does not include the metabolite cocaine N-oxide that has been detected in rat and human plasma.
Cocaine is a strong hepatotoxin in mice, producing fatty infiltration, midzonal and periportal necrosis and elevation of serum glutamic oxaloacetic transaminase (SGOT). The mechanism of cocaine toxicity to liver is not well understood but appears to be associated with actions of one or more of N-oxidative metabolites of cocaine (Shuster et al., 1977).
In this study, molecular modelling analyses have been carried out using the programs HyperChem 7.0 (HyperChem, 2002) and Spartan ’02 (Spartan, 2002) to investigate the relative stability of cocaine and its metabolites. In an earlier study, Paula et al. (2004) investigated 3D structure-activity relationship of the cocaine binding by a novel human monoclonal antibody (Perino et al., 1978).
COMPUTATIONAL METHODS
The geometries of cocaine, cocaethylene androecgonine methyl ester androecgonine, norcocaine, ecgonine methyl ester, ecgonine, benzoylecgonine and cocaine N-oxide have been optimized based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling programs Spartan ’02 and HyperChem 7.0. Molecular mechanics calculations were carried out using MM+ force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried using the program Spartan ’02 at B3LYP/6-31G* level. The order of calculations: molecular mechanics followed by semi-empirical followed by DFT minimized the chances of the structures being trapped in local minima rather reaching global minima. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structures corresponding to global minimum or close to that were reached in most cases. Although RMS gradient of 0.001 may not be sufficiently small for vibrational analysis, it is believed to be sufficiently low for calculations associated with electronic energy levels.
In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimized structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment and solvation energy, HOMO and LUMO. The research was done in the School of Biomedical Sciences, The University of Sydney during the period July 2005 to March 2006.
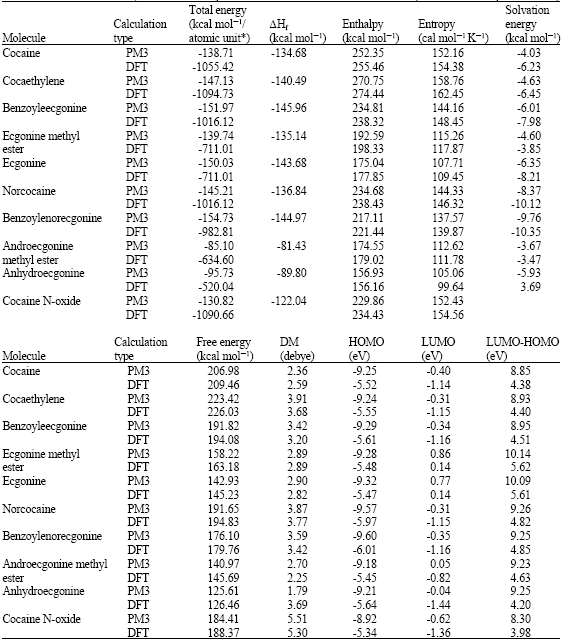
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for cocaine, cocaethylene, benzoylecgonine, ecgonine methyl ester, ecgonine, norcocaine, benzoylenorecgonine androecgonine methyl ester androecgonine and cocaine N-oxide. Figure 2-11 give the optimized structures for cocaine, cocaethylene, benzoylecgonine, ecgonine methyl ester, ecgonine, norcocaine, benzoylenorecgonine androecgonine methyl ester androecgonine and cocaine N-oxide as per PM3 calculations using the program HyperChem 7.0. The structures also give (a) 2D contours of total electrostatic potential and (b) HOMOs. The dotted arrows indicate the positions of most negative electrostatic potential in (a) and HOMOs with the highest electron density in (b).
| Table 1: | Calculated thermodynamic and other parameters for cocaine and its metabolites (DM stands for dipole moment) |
 | |
| * In atomic unit from DFT calculations | |
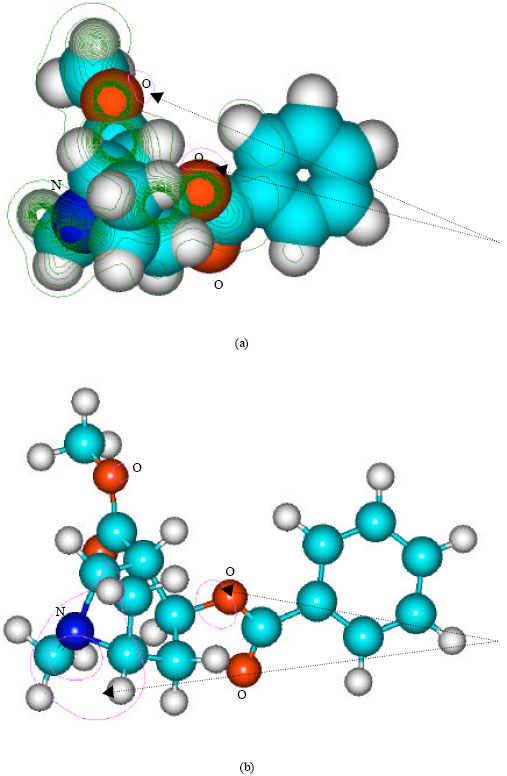 | |
| Fig. 2: | Structure of cocaine giving (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
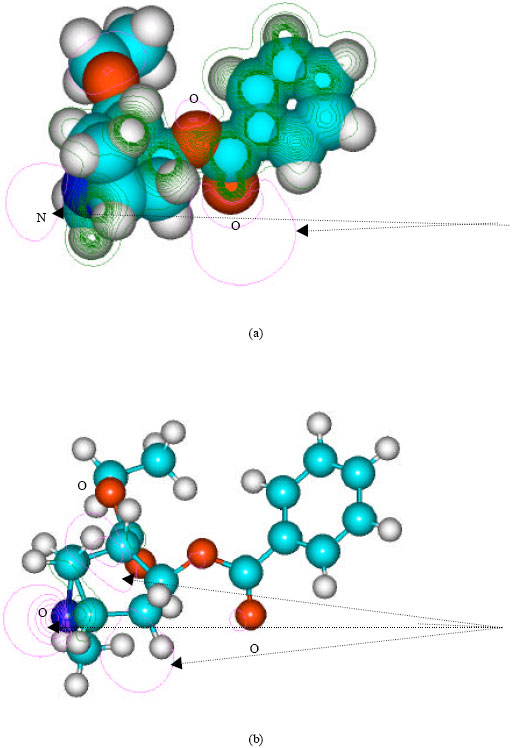 | |
| Fig. 3: | Structure of cocaethylene giving (a) 2D contours of total electrostatic potential and (b) 2D HOMO plot |
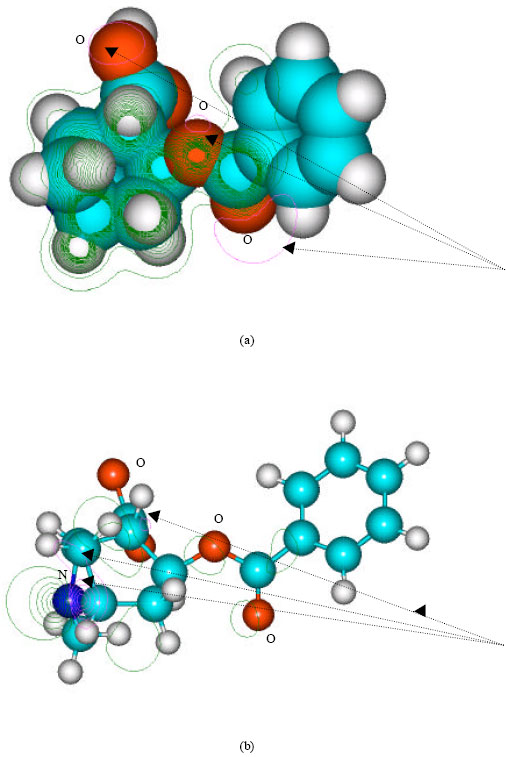 | |
| Fig. 4: | Structure of benzoylecgonine giving (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
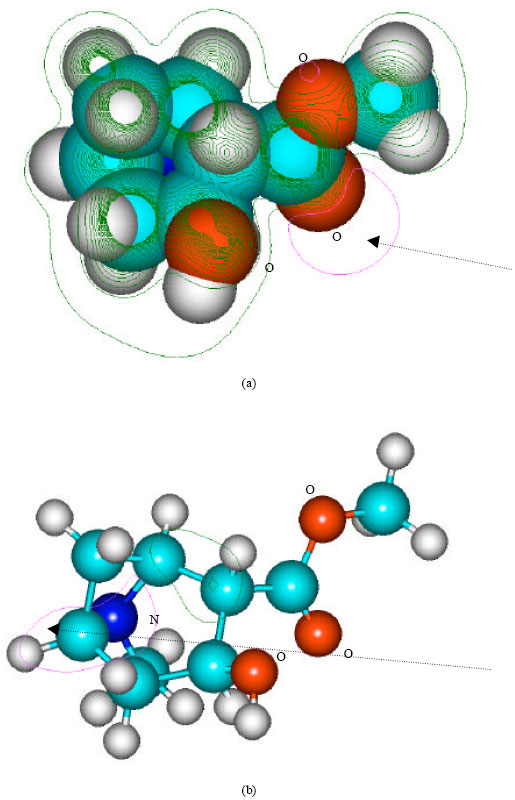 | |
| Fig. 5: | Structure of ecgonine methylester giving (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
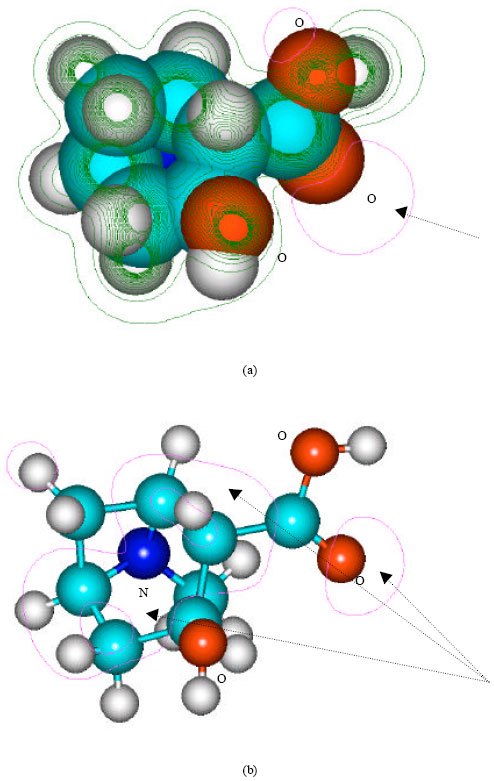 | |
| Fig. 6: | Structure of ecgnonine giving (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
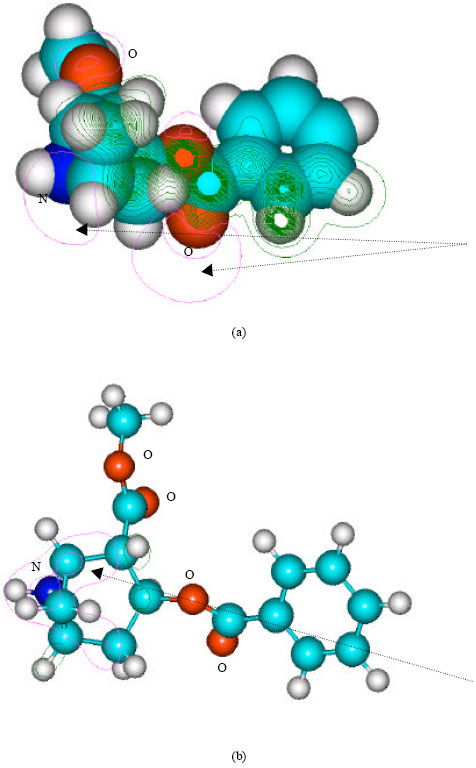 | |
| Fig. 7: | Structure of norcocaine giving (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
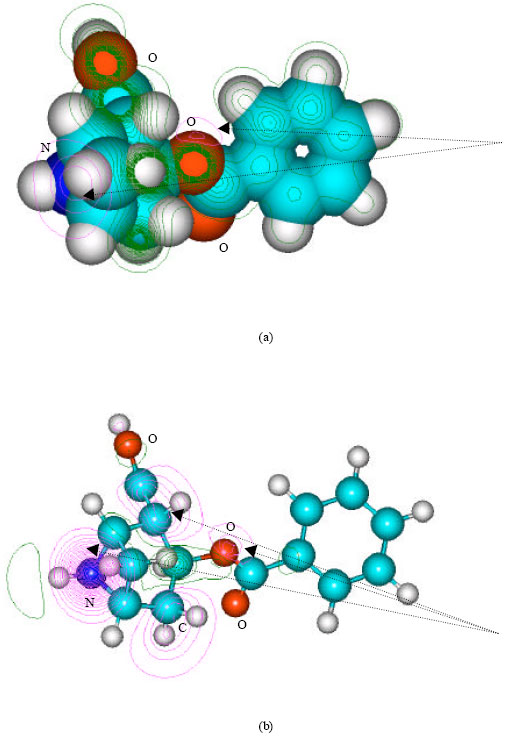 | |
| Fig. 8: | Structure of benzoylnorecgonine giving (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
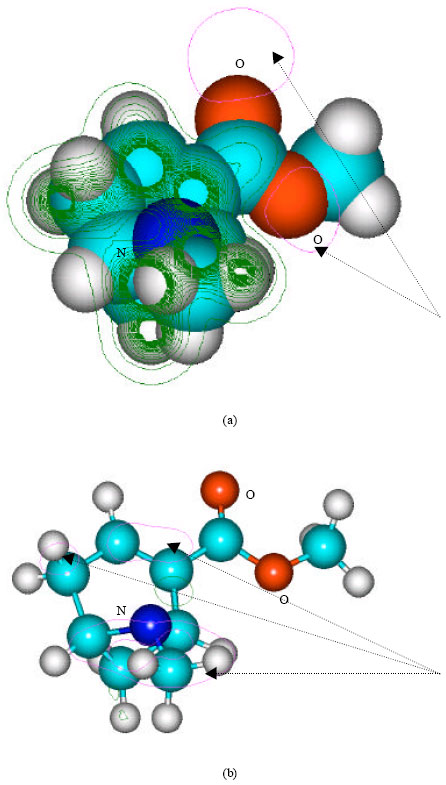 | |
| Fig. 9: | Structure of androecgonine (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
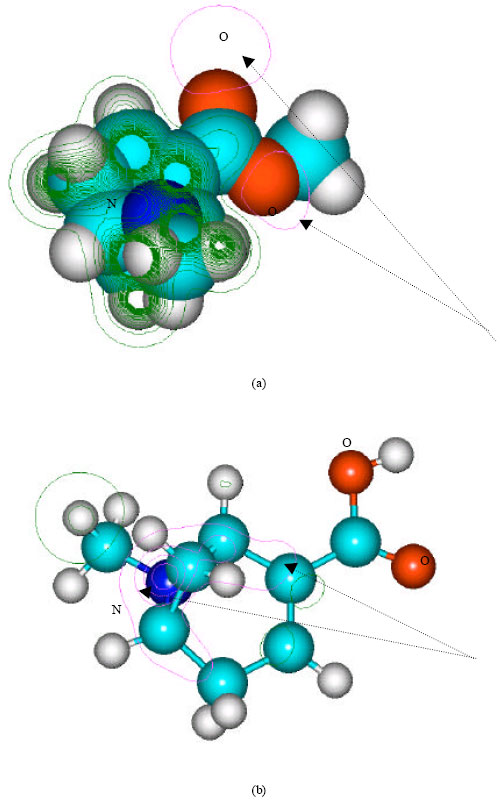 | |
| Fig. 10: | Structure of anhydroecgonine giving 2D contours of total electrostatic potential showing (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
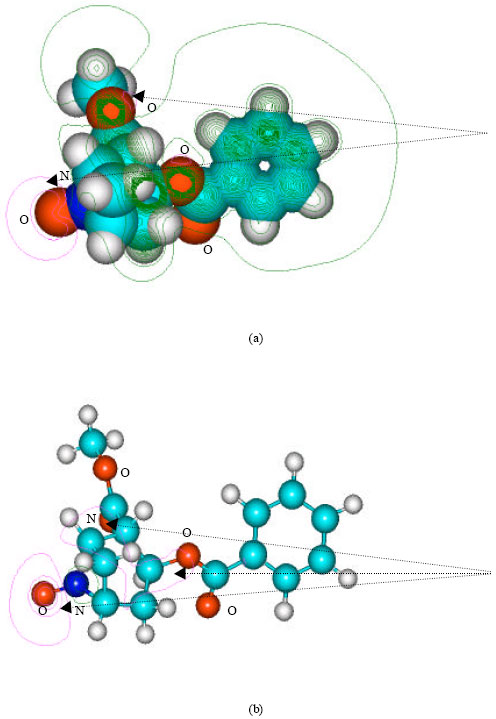 | |
| Fig. 11: | Structure of cocaine N-oxide giving 2D contours of total electrostatic potential showing (a) 2D contours of total electrostatic potential and (b) 2D plot of HOMO |
Among the metabolites of cocaine, norcocaine, ecgonine, benzoylenorecgonine and benzoylecgonine have large energies of solvation, indicating the compounds would be highly soluble in water and hence can be readily excreted via urine. The parent compound and the other metabolites also have reasonably large solvation energies so that they can also be appreciably soluble in water. The hydrochloride forms of the compounds would have much higher solubility in water. The solvation energy of cocaine N-oxide could not be calculated by Spartan’02 as it contained an unpaired electron. As expected, the solvation energy of benzoylecgonine formed from the hydrolysis of cocaine in which a methyl group is replaced by a hydrogen atom, is found to be greater than that of cocaine (-7.98 kcal mol-1 for the former as against -6.23 kcal mol-1 for the latter as per DFT calculations). Similarly, the solvation energy of ecgonine is found to be greater than that of ecgonine methyl ester (-8.21 kcal mol-1 for the former as against -3.85 kcal mol-1 for the latter from DFT calculations). Likewise, the solvation energy of anhydroecgonine is found to be greater than that of anhydroecgonine methyl ester.
It can be seen that in the case of cocaine, the positions of more negative electrostatic potential are centered on oxygens of the ester linkages whereas the HOMOs with the high electron densities are found to be centered on the oxygen atom of the ester bond that connects the aromatic and the heterocyclic rings and the tertiary nitrogen atom. The very high negative electrostatic potential around oxygens of the ester linkages indicates that the positions would be more susceptible to electrophilic attack.
In the case of cocaethylene, the positions of more negative electrostatic potential are centered on oxygen of the ester linkage that connects the aromatic ring with the heterocyclic ring containing the tertiary nitrogen atom, the carbonyl oxygen of the same ester group and the tertiary nitrogen atom whereas the HOMO with the highest electron density is centered on the tertiary nitrogen. The very high negative electrostatic potential around oxygens of the ester linkages and the tertiary nitrogen indicates that the positions would be susceptible to electrophilic attack.
In the case of benzoylecgonine, the positions of most negative electrostatic potentials are found to be centered on the hydroxyl and carbonyl oxygens and oxygen of the ester linkage whereas the HOMO with the highest electron density is found to be close to the tertiary nitrogen atom. In the case of ecgonine methylester, ecgnonine, benzoylnorecgonine and anhydroecgonine, the positions of negative electrostatic potential are centered mostly on the oxygens. In the case of cocaine N-oxide, the position of most negative electrostatic potential is found to be centered on the oxide oxygen as is the HOMO with the highest electron density, indicating that the position would be highly susceptible to electrophilic attack.
When the LUMO-HOMO energy differences of cocaine and its metabolites are compared, it is found that most of them have values ranging from 5.4 to 5.6 eV as per DFT calculations except nicotine N-oxide which has a smaller value of 3.98 eV and anhydroecgonine which has a value of 4.20 eV. The values suggest that whereas cocaine and most of its metabolites have similar kinetic lability, anhydroecgonine and cocaine N-oxide have greater kinetic lability-the most labile compound being cocaine N-oxide. It should be noted that only recently cocaine N-oxide has been identified as a metabolite of cocaine whose concentration in blood plasma is found to be quite low. Gas chromatography/mass spectrometry (GC/MS) is often used to detect and quantify cocaine metabolites in biological specimens. Cocaine N-oxide being thermally unstable may decompose in the GC/MS so that the measured value of the metabolite may not reflect the true value produced from the metabolism of cocaine. The higher reactivity of cocaine N-oxide explains why the metabolite is thermally unstable and can be expected to be highly toxic-cocaine N-oxide may bind with glutathione, metalothionein and cysteine so as to alter the anti-oxidant status of the cell. The density of electrostatic potential on the surface of cocaine N-oxide given in Fig. 12 shows that the surface of the metabolite abounds in electron-deficient (blue) regions which make it susceptible to nucleophilic attack e.g., by reduced form of glutathione and nucleobases in DNA.
 | |
| Fig. 12: | Density of electrostatic potential on the surface of cocaine-N-oxide showing abundance of blue electron-deficient regions |
The high reactivity and ability to cause glutathione depletion and cause oxidation of nucleobases in DNA mean that cocaine N-oxide can induce cellular toxicity and cause damage to DNA.
Molecular modelling analyses show that the most labile metabolite of cocaine, namely cocaine N-oxide has the smallest LUMO-HOMO energy difference and hence greatest kinetic lability. The surface of the metabolite is also found to abound in electron-deficient regions so that it can cause oxidation of reduced form of glutathione and that of nucleobases in DNA, thus compromising the antioxidant status of the cell and inducing damage to DNA.