Research Article
Molecular Modelling Analysis of the Metabolism of Niclosamide
School of Biomedical Sciences, Faculty of Health Sciences, The University of Sydney, Australia
Niclosamide (2’,5-dichloro-4’-nitrosalicylanilide) is a restricted-use pesticide that has been successfully used alone and in combination with 3-trifluoromethyl-4-nitrophenol (TFMNP) for more than 40 years to control sea lampreys (Petromyzon marinus) in streams tributary to the Great Lakes (Hubert et al., 2005). A single seal lamprey is capable of killing as much as 18 kg of fish during a period of 12-20 months of its adult life (Graebing and Chib 2004). As the chemical is generally applied directly to waterways inhabited by fish, there is a potential for niclosamide residue to accumulate in fish muscle tissue. Niclosamide is also used to kill golden apple snail (Pomacea canaliculata) which is a major pest of rice. Golden apple snail was introduced in Philippines as an alternative protein source but soon became a pest due to its rapid breeding rate and voracious feeding on rice seedlings (Calumpang et al., 1995). The primary action of niclosamide appears to involve inhibition of energy production in mitochondria of parasites through inhibition of anaerobic phosphorylation of adenosine diphosphate (ADP). The drug is effective against adult tape worms but not against ova (Winter, 1992).
Despite its general use, niclosamide is found to be toxic to several aquatic organisms (Lardans and Dissous, 1998; Andrews et al., 1982). Long administration has also been found to cause problems to terrestrial and aquatic plants (Lardans and Dissous, 1998). Niclosamide has been found to bind with DNA after its reductive activation that may be the cause of its toxicity (Abreu et al., 2002).
Niclosamide is resistant to hydrolysis at pH 5, 7 and 9 for over 4 months (El-Dib and Aly, 1976) but photolysis of the 70% wettable powder formulation in aqueous solutions results into a significant decrease in biological activity within 24 h (Gillet and Bruau, 1961; Statham and Lech, 1975).
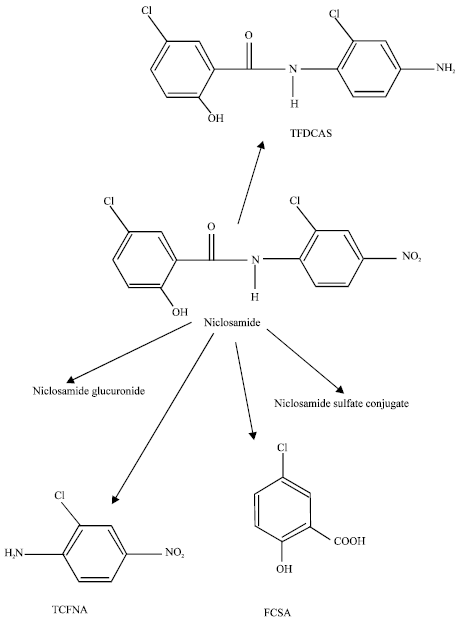
Study on the metabolism of niclosamide demonstrated that niclosamide-glucuronide is the major metabolite of niclosamide in the bile of rainbow trout (Graebing and Chib, 2004). Niclosamide residues in fish begin to decline 12-17 h after exposure as the compound is metabolized to niclosamide glucuronide and eliminated (Griffiths and Facchini, 1979).
 | |
| Fig. 1: | Proposed metabolic pathways for niclosamide and its metabolites (Rotzinger et al., 1999) |
Phase I metabolism of niclosamide may result in a hydrolytic cleavage of the amide bond to produce 5-chlorosalicylic acid (FCSA) and 2-chloro-4-niroaniline (TCFNA) as the main metabolites (Fig. 1) (Rotzinger et al., 1999). Another minor metabolite is 2’,5-dichloro-4’-aminosalicylanilide (TFDCAS). Bacterial mutagenicity tests with these two compounds reveal that FCSA is not mutagenic but TCFNA is and at a lower level than niclosamide (Espinosa-Aguirre et al., 1991). Additional results from genotoxic studies in rodents and humans suggest that niclosamide is absorbed from the gastrointestinal tract and mutagenic metabolites are excreted in the free form and also as glucuronides.
In this study, molecular modelling analyses have been carried out using the programs Hyper Chem 7.0 (Hyper Chem, 2002) and Spartan ’02 (Spartan, 2002) to investigate the relative stability of niclosamide and its metabolites with the aim of providing a better understanding on the relative toxicity due to niclosamide and its metabolites.
The geometries of niclosamide and its metabolites FCSA, TCFNA and TFDCAS have been optimised based on molecular mechanics, semi-empirical and DFT calculations, using the molecular modelling programs HyperChem 7.0 and Spartan ’02. No calculations were done for the glucuronides of the compounds. Molecular mechanics calculations were carried out using MM+ force field. Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried using the program Spartan ’02 at B3LYP/6-31G* level. In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimised structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for HOMO and LUMO. The order of calculations: molecular mechanics followed by semi-empirical followed by DFT minimized the chances of the structures being trapped in local minima rather than reaching global minima. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structures corresponding to global minimum or close to that were reached in most cases. Although RMS gradient of 0.001 may not be sufficiently small for vibrational analysis, it is believed to be so for calculations associated with electronic energy levels. For the optimised structures, single point calculations were carried to give heat of formation, enthalpy, entropy, free energy, surface area, volume, dipole moment and solvation energy, HOMO and LUMO. The work was carried out in the School of Biomedical Sciences, The University of Sydney during November 2005 to April 2006.
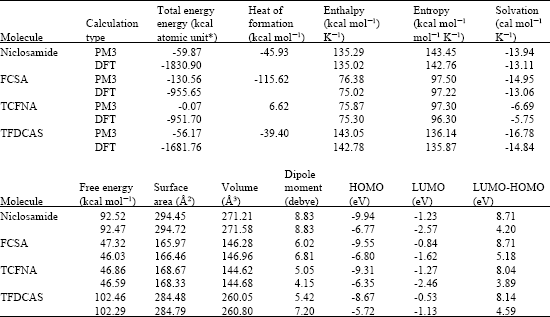
Table 1 gives the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, surface area, volume, solvation energy, dipole moment, energies of HOMO and LUMO as per both PM3 and DFT calculations for niclosamide and its metabolites FCSA, TCFNA and TFDCAS. (where red indicates HOMOs with high electron density) in (b), LUMOs in (c) and surface charges (where red indicates negative, blue indicates positive and green indicates neutral) in (d) as applied to the optimised structures of niclosamide and its metabolites FCSA, TCFNA and TFDCAS.
| Table 1: | Calculated thermodynamic and other parameters of niclosamide and its metabolites |
 | |
| * in atomic units from DFT calculations | |
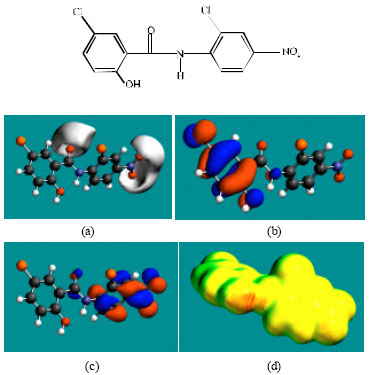 | |
| Fig. 2: | Structure of niclosamide giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
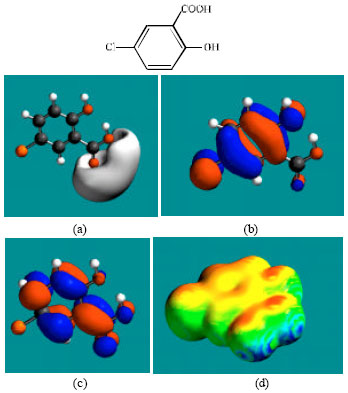 | |
| Fig. 3: | Structure of FCSA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
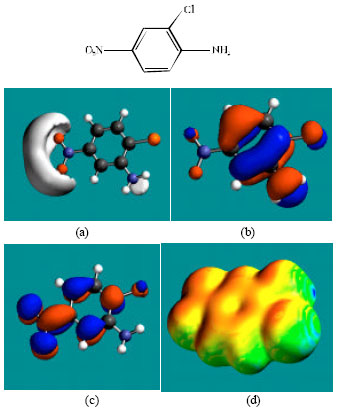 | |
| Fig. 4: | Structure of TCFNA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
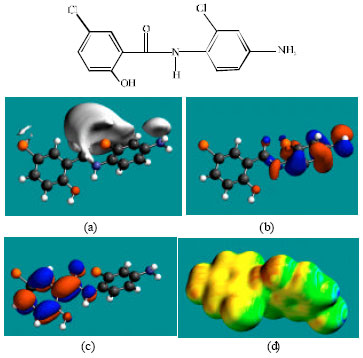 | |
| Fig. 5: | Structure of TFDCAS giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) surface electric charges (where red indicates negative, blue indicates positive and green indicates neutral) |
The calculated solvation energies of niclosamide and its metabolites FCSA, TCFNA and TFDCAS from PM3 calculations in kcal mol-1 are respectively -13.94, -14.95, -6.69 and -16.78, respectively and their dipole moments from DFT calculations are 8.83, 6.81, 4.15 and 7.20, respectively. The results indicate that niclosamide and its metabolites FCSA and TFDCAS would have similar solubility in Figure 2-5 give the regions of negative electrostatic potential (greyish-white envelopes) in (a), HOMOs water whereas the other metabolite TCFNA would have a much lower solubility in water. The lower solubility of TCFNA in water means that it will be less easily excreted in the urine. It will however be seen later that TCFNA would be most labile kinetically and hence would react most readily with biomolecules.
Niclosamide and its metabolites vary significantly in their LUMO-HOMO energy differences (that range from 3.89 eV to 5.18 eV as per DFT calculations) so that they would vary in their kinetic lability. TCFNA has the smallest LUMO-HOMO energy difference (3.89 eV from DFT calculations) so that (as noted earlier) the metabolite would be most kinetically labile. This may make TCFNA as being the most toxic metabolite as it will react with biomolecules more readily and may also explain why TCFNA is highly mutagenic.
When heat of formation values are compared, it is found that FCSA has a more negative heat of formation than the parent drug niclosamide (-115.62 kcal mol-1 for the former and -45.93 kcal mol-1 for the latter) but TCFNA has a positive heat of formation (+6.62 kcal mol-1). The values suggest that FCSA may be more stable and TCFNA less stable than niclosamide (and if so TCFNA would be unstable thermodynamically as well).
In the case of niclosamide, the electrostatic potential is found to be more negative around the carbonyl oxygen atom and the oxygen atoms of the nitro group, indicating that the positions may be subject to electrophilic attack. In the case of FCSA, the electrostatic potential is found to be more negative around the carboxyl and hydroxyl oxygen atoms, indicating that the positions may be subject to electrophilic attack. In the case of TCFNA, the electrostatic potential is found to be more negative around the nitro group and the amino nitrogen atom, indicating that the positions may be subject to electrophilic attack. In the case of TFDCAS, the electrostatic potential is found to be more negative around the amino group of the aniline ring, the linking amino nitrogen and the chlorine atoms, indicating once again that the positions may be subject to electrophilic attack.
In the case of niclosamide and TFDCAS, the HOMOs with high electron density are found above and below the phenolic ring whereas LUMOs are found close to the atoms of the nitro-substituted phenyl ring. In the case of FCSA and TCFNA, the HOMOs and LUMOs with high electron density are found above and below the atoms of the phenyl ring and the hydroxyl oxygen atom.
The abundance of yellow regions with some green and red patches on the surfaces of niclosamide, FCSA and TCFNA indicates the surfaces may have an overall negative charge. It also means that the interaction of niclosamide, FCSA and TCFNA with biomolecules including water is more likely to be hydrophilic rather than hydrophobic so that the molecules would be expected to be to some extent soluble in water. The surfaces of FCSA and TCFNA have more of red regions than that of niclosamide so that the surface of these compounds would be more negatively charged.
Molecular modelling analyses show that niclosamide and its metabolites differ in their kinetic lability, solubility in water and possibly in their thermodynamic stability. TCFNA has the smallest LUMO-HOMO energy difference and hence it would be most labile kinetically. Also, it has the smallest solvation energy so that it will be least soluble in water and hence least easily excreted in the urine. Higher kinetic lability, lower excretion rate and possibly lower thermodynamic stability may make TCFNA highly toxic and mutagenic.
Fazlul Huq is grateful to the School of Biomedical Sciences, The University of Sydney for the time release from teaching.
mostafa abdelrahman Reply
very good thank you