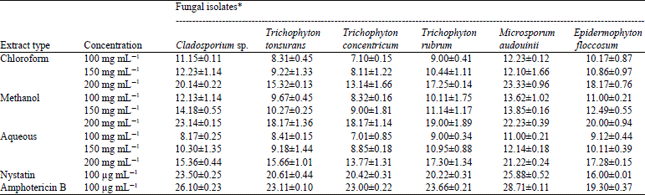
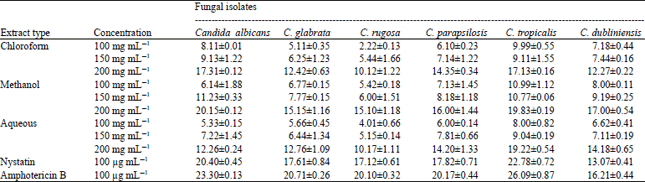
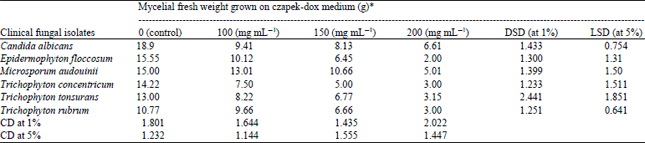
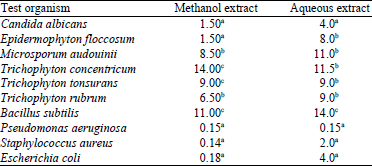
Research Article
An Evaluation of the Antimicrobial Activity of Commiphora myrrha Nees (Engl.) Oleo-gum Resins from Saudi Arabia
Department of Clinical Laboratory Sciences, College of Applied Medical Sciences, Shaqra University, Saudi Arabia
A.M. Saadabi
Department of Clinical Laboratory Sciences, College of Applied Medical Sciences, Shaqra University, Saudi Arabia
Mohammed I. Alghonaim
Department of Biology, College of Science and Humanities, Al-Quway`iyah, Shaqra University, Saudi Arabia
Khalid Elfakki Ibrahim
Department of Zoology, College of Science, King Saud University, Riyadh, Saudi Arabia