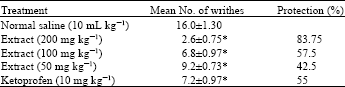Research Article
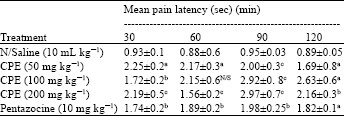
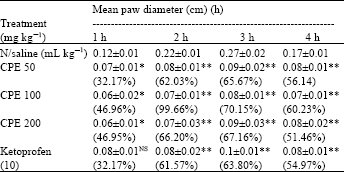
Phytochemical Screening and Evaluation of Analgesic and Anti-inflammatory Activities of the Methanol Leaf Extract of Cissus polyantha
Department of Pharmaceutical and Medicinal Chemistry, Ahmadu Bello University, Zaria, Nigeria
A.M. Musa
Department of Pharmaceutical and Medicinal Chemistry, Ahmadu Bello University, Zaria, Nigeria
A.H. Yaro
Department of Pharmacology, Faculty of Medicine, Bayero University, Kano, Nigeria
M.B. Sani
Department of Pharmacology and Therapeutics, Ahmadu Bello University, Zaria, Nigeria
A. Amoley
Faculty of Pharmaceutical Sciences, Ahmadu Bello University, Zaria, Nigeria
M.G. Magaji
Department of Pharmacology and Therapeutics, Ahmadu Bello University, Zaria, Nigeria