ABSTRACT
Frequency of contamination in home slaughtered meat Jimma town south west Ethiopia was examined in the present study. The microbiological safety and quality issue is the pro-active measure to prepare the meat for consumption. A total 48 raw meat samples including beef (n = 4), Mutton (n = 37), chevron (n = 7) were collected from individual household and small food seller house in the town were analyzed for microbiological contamination using conventional culture methods. The bacterial spp. isolated from the samples were Bacillus, Micrococcus species, Salmonella species, Enterobacteriacea, coliform, Staphylococcus species and other gram positive rods, some of which harbor human pathogens of public health concern. The aerobic mesophilic counts varied from 0.19 to 3.67 log10 CFU g-1. Coliforms were encountered in all samples. Staphylococci were present at 0.95-2.28 log10 CFU g-1 in 36% (8) mutton and 50% (2) chevron, samples. Salmonella species were present in 10.42% (5.0) of all the samples. The majority of meat samples investigated in this study had contaminant microorganism and some pathogens. The present study revealed that despite very low contamination rate, foodstuffs particularly beef, chevon and mutton parts could be a potential vehicle for foodborne infections and implementation of preventive measures and consumer food safety education efforts are needed.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jms.2013.779.784
URL: https://scialert.net/abstract/?doi=jms.2013.779.784
INTRODUCTION
The big and small ruminant’s animals (cattle, sheep and goats) are reared all over Ethiopia. The high production of animals creates advantage in employment, income generation and food (beef, chevon and mutton). Bekele and Butako (2010) reported that cattle, sheep and goats are slaughtered mostly during Christmas, Islamic festivals and months prior to the farming season. Beef, chevon and mutton are therefore popular meats in Ethiopia, which are cherished by most Ethiopian (Dabassa and Bacha, 2012). These meats are normally cut into pieces, weighed in money and then sold in restaurants to consumers and also used by the individual at their home.
The risk of the transmission of zoonotic infections is also associated with contaminated meat (Ali et al., 2010). The common route for food borne illness and their consequence always was involving through their transmission to human during production, handling and consumption of meat and meat products (Callaway et al., 2008). However, a few community-based reports provide evidence of several outbreak caused by Salmonella, Shigella, E. coli and Listeria spp. in different parts of the world depending on a range of factors including the organism, geographical factors, farming and/or meat production practices (Iroha et al., 2011). Animal diseases that can be transmitted to humans have to be controlled at all stages from farm to fork for its risk and also for the customer’s attitude, behavior and benefit (Callaway et al., 2008).
Although, beef, chevon and mutton play a significant role in the livelihood of most Ethiopian, they can be important source of food-borne human pathogens and subsequently food-borne diseases due to the poor handling conditions of meat in most Ethiopian markets (Dabassa and Bacha, 2012). In addition meat which is rich in protein is a good medium for growth of microbes (Bacha et al., 2007; Zakpaa et al., 2009; Ukut et al., 2010). Bacha et al. (2007) indicated that, meat is the best growth media for the spoilage microorganisms, as the results of the nature of the food by itself, which means the nutritional and water content of the meat. As the results of absence of HACCP guideline in the country like Ethiopia, the awareness on microbiological safety and quality of meat in the community is crucial in relation to the trends of meat processing at home. This study was therefore undertaken to assess the level of microbial contamination in beef, chevron and mutton slaughtered at home and used for consumption by the community in Jimma town South Western Ethiopia.
MATERIALS AND METHODS
Study area and period: The study was conducted in Oromia regional state, Jimma zone, Jimma town from October 2012 to march 2013. Jimma town is located at 353 km to the south west from the capital of Addis Ababa (Fig. 1).
Sample collection: A total of 48 carcass samples were collected from household and small food selling house located in Jimma town between October, 2012-March, 2013. These consisted of 4 carcass samples of beef, 37 samples of Mutton and 7 samples of Chevron. All samples were collected by the person dressing the carcass aseptically and immediately brought to the microbiology laboratory of Collage of Natural Sciences, Jimma University for microbiological analysis. Microbiological analysis was conducted within 1 to 3 h of sample collected.
Microbiological analysis: To test for the presence of Microorganisms, 25 g of carcasses samples were separately mixed with 225 mL Peptone Water (PW), homogenized for 15 min and appropriate serial dilution were done.
Microbial enumeration: From appropriate serial dilutions, 0.1 mL aliquots were spread-plated in duplicate on pre-dried surface of Plate count Agar (oxoid) plates, mannitol Salt agar (Oxoid) plates, Violet Red Bile glucose Agar (oxoid) plates, Violet Red Bile Agar (oxoid) plates, Chloramphenicol Bromophenol Blue Agar. Colonies were counted after the culture media were incubated at 37°C for 24 h, for isolation of aerobic mesophilic bacteria, Micrococcus, Staphylococcus, Enterobacteriacea, Coli forms and yeast and molds.
Salmonella species: For the isolation of Salmonella species, the samples were pre-enriched in buffered peptone water (incubated aerobically at 37°C for 24 h), followed by secondary enrichment in rappaport vassiliadis broth (incubated aerobically at 37°C for 24 h) and plating onto xylose lysine deoxycholate agar (incubated aerobically at 37°C for 24 h).
Yeast and molds: For the isolation of Yeast and molds, from appropriate dilution, 0.1 mL aliquots were spread-plated in duplicates on pre-dried surfaces of Chloramphenicol Bromophenol Blue Agar made from the following ingredients: yeast extract, 5 g; glucose, 20 g; Chloramphenicol, 0.1 g; Bromophenol blue, 0.01 g; agar, 15 g; distilled water, 1000 mL; pH, 6-6.4.
 | |
| Fig. 1: | Study area map |
The seeded culture plates were incubated at 25-28°C for three to five days. Smooth (non-hairy) colonies without extension at periphery (margin) were counted as yeast.
Flora analysis: After enumeration of aerobic mesophilic bacteria, about 10-20 colonies were picked randomly from countable plates and inoculated into tubes containing about 5 mL Nutrient broth (oxoid). These were incubated at 30-35°C over night. Cultures were purified by repeated plating and were characterized to the genus level and various bacterial groups using the following tests.
Cell morphology: From overnight pure broth culture, gram stain and hanging drop was prepared on microscope slide. The preparation was observed under the microscope. The morphological criteria considered during the observation were: Cell Shape: Regular: rods, or cocci forms; Irregular: branched, coryneforms, pleomorph; Cell arrangement: Single, pairs, clusters, chains and tetrads; Motility: Motile, non-motile and Endospore: Present, absent.
Identification and confirmation of bacterial isolates: The bacteria colonies obtained were purified on nutrient agar. They were then identified and confirmed using Gram staining and biochemical tests such as motility, catalase, coagulase, indole, citrate utilisation, oxidase, urease hydrolysis Triple Sugar Iron (TSI agar) utilization, SIM medium Test, Lysine iron agar Test, fermentation of glucose, sucrose and Manitol, Metyl red-Voges proskauer.
Serological test: The suspected Salmonella was confirmed with serological test according to Gruenewald et al. (1990) methods: Polyvalent Flagellar (H) Test: From the appropriate culture, Add 2.5 mL formalinized saline solution to the broth culture. Select two formalinized broth cultures and test with Salmonella polyvalent flagellar (H) antisera. Add 0.5 mL of formalinized culture to 0.5 mL of polyvalent flagellar (H) antiserum in a small test tube (10x75 mm). Prepare saline control by mixing 0.5 mL formalinized saline with 0.5 mL antiserum. Incubate mixtures in a 48-50°C water bath and observe agglutination at 15 min. intervals. Read final results after one hr.
Polyvalent somatic (O) test: Using a wax pencil mark off test and control sections (about 1 cm square) on a glass slide. Prepare a heavy suspension by emulsifying a loopful of culture from the presumed-positive TSI slant in 1 mL saline solution. Place 1 drops of the polyvalent O antiserum on the test section and 1 drop of the saline solution on the control section. Transfer a loopful of culture suspension to the saline drop. Flame the loop and transfer a second loopful of the suspension to the antiserum section. Tilt the slide in a back-and-forth motion for 1 min. Read the slide and consider any degree of agglutination as a positive reaction (Gruenewald et al., 1990).
RESULTS AND DISCUSSION
In Jimma almost all individual users and small food selling house were use home slaughtered meat. Most of the time, sheep and goats are slaughtered at home and beef were in rare case. The eviscerations were done by the specialist with a payment by the owner of food seller house and the individuals. A total of 397 bacteria were isolated and the frequency of the isolated bacteria from different meat samples was summarized in (Table 1) as follows; 27.2 % of B. cereus was isolated from all the meat sampled while 21 and 19 isolates were from mutton and chevron, respectively.
Furthermore, 5.04% of E. coli was isolated while 9 isolates were from beef, 8 from mutton and 5 from chevron and 4.2% of klebsella was isolated and 6 isolates were from beef, 3 from mutton and 1 from chevron, 1.26% of salmonella isolated , 3 isolates were from beef, 1 from mutton and 1 from chevron, 9.32% of proteus was isolated, 14 isolates were from beef, 8 from mutton and 11 from chevron, 9.57% of Staphylococcus spp. was isolated, 19 isolates were from beef 9 and 10 each from mutton and chevron, respectively. Significant variation were noted in the counts of most microbial groups among meat samples (CV = 1.36-66.35%) (Table 1).
It was shown in this study that the predominant bacteria isolated were Bacillus spp, Salmonella, Staphylococcus spp and Klebsiella spp, Micrococcus, Proteus and E. coli (Table 1 and 2) and yeasts and molds. The total aerobic bacterial count varied with the type of animal and from sample to sample (Table 2). The total bacterial count for beef ranges from log 0.06-log 3.67 CFU g-1, chevon ranged from log 0.19-log 1.72 CFU g-1 and that of mutton was log 0.48-log 2.62 CFU g-1. Beef had the highest aerobic Mesophylic bacteria count of log 3.67 cfu g-1 followed by mutton with log 2.62 cfu g-1.
Generally, 39.79% (158) of the meat sample were dominated by gram positive organisms. Aerobic mesophilic (30.98%) and Bacillus spp. (27.2%) were the dominating bacterial flora in meat samples followed by gram positive Staphylococci families (38%) (Table 1).
| Table 1: | Bacteria spp. isolated from carcasses in small food sell house and individual houses slaughterhouse |
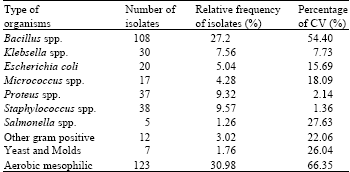 | |
| CV%, percentage of coefficient of variation | |
| Table 2: | Microbial counts (log CFU g-1) and average mean colony forming unit of meat samples collected from individual household and small food selling house in Jimma town |
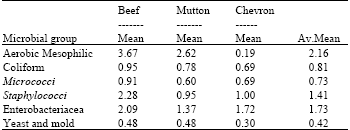 | |
Micrococcus, Enterobacteriacea and coli forms were also encountered in all meat samples.
Cattle carcasses, which are initially sterile, become contaminated with bacterial pathogens via transmission of organisms from the exterior of the live animal and/or from the environment, to the meat surface. Extensive contamination, or abusive conditions that allow bacteria to reproduce, increase risk for presence of pathogenic bacteria and formation of toxins in food. Fresh raw meat like Beef, Mutton and Chevon has been implicated for number of meat borne infections and intoxications in several countries (Mukhopadhyay et al., 2009). The high level of bacterial viable counts after post washing of the carcasses in this study is in agreement with the study of Ali et al. (2010) who recorded that the highest contamination was at the point of washing, on different sites of examination of bovine carcasses. Another study by Jeffery et al. (2003) revealed that the workers hands and the equipment were the sources of meat contamination and these results are in accord with the present results. The elimination of contamination sources by practicing good sanitary measures will reduce the occurrence of microorganisms. Appropriate methods should be applied during slaughtering operations, using adequate water and disinfection. Such control measures should include an extensive education programs for proper hygiene and improvement of managements.
| Table 3: | Frequency distribution of the isolated bacteria from different the meat samples examined during this study |
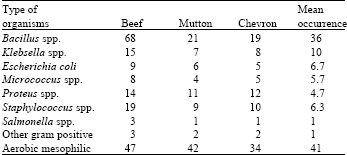 | |
Micrococcus were also encountered in more than 4.28% of different meat samples with counts <log0.91 CFU g-1 in most meat samples. On contrary, examination of the surface of freshly slaughtered beef (Kitcheil, 1962) also showed the high prevalence (50%) of Micrococcus. From among the different samples collected from various places around the slaughterhouse, Kitcheil (1962) isolated 18 different species of micrococci and estimated that they constituted about 46-47% of the micro-flora of the slaughterhouse environment.
Bacillus spp and aerobic mesophyilic bacteria were the dominant microorganisms in the three meat group (Table 3). Stataphylococus spp and Proteus are the third and the fourth Bacterial spp., respectively. Similarly the microbial contamination beef were greater than the mutton and chevron. These indicates that the size of an animal supports to contact with different bodies and make more contamination by microorganisms.
The bacterial counts for the meat samples were generally low although they were below 107 where spoilage of meat occur (Nolan et al., 1980). Thus, meats slaughtered at home in Jimma town probably had a low microbial load. Nevertheless the isolation of Salmonella spp., Staphylococcus spp., Bacillus spp., E. coli and Molds can be worrying because certain strain of these bacteria and fungus causes food-borne infections and intoxication. Salmonella, Staphylococcus, Bacillus spp., E. coli and mold infections can be contracted through consumption of contaminated beef, chevon and mutton. Salmonellas are important causes of gastroenteritis (Callaway et al., 2008). Staphylococcus spp. can be part of normal flora on the skin of humans and animals which can be transmitted from person to product through unhygienic practices (Raji, 2006). Staphylococcus spp. cause infections such as arthritis, black pox, boil, bronchitis, bumble foot, carbuncle, cystitis, endocarditis, meningitis, osteomyelitis, pneumonia and scalded skin (Adzitey et al., 2010). Mould can produce toxic substances known as mycotoxins. Ingesting aflatoxin can cause liver cancer. Coliforms were encountered in all samples analyzed (Ogiehor et al., 2007). Escherichia coli causes illness ranging from gastrointestinal tract-related complications such as diarrhea, dysentery, urinary tract infection, pneumonia and even meningitis (Adzitey et al., 2010), although majority of the Escherichia coli strains are non-pathogenic and exist in the intestinal tract of humans and animals.
The presence of the isolated bacteria can be traced back to the handling and processing methods of meat in the Jimma (Ethiopia). Animals are slaughtered at home and restaurant without observing strict hygienic practices. It is also a common practice to see people carrying carcasses just after dressing on their bare with out garment. The meats were stored in unsafe condition. Chevron and Mutton contamination by various microorganisms have also been reported in other countries by different authors. Selvan et al. (2007) found that the mean total viable count was significantly greater in mutton products than all other products (chicken, pork and beef) studied. Mukhopadhyay et al. (2009) tested 23 chevon samples, out of which five samples (21.74%) yielded Staphylococcus aureus. In addition Sharma et al. (1993) also identified Staphylococcus aureus, E. coli, Bacillus sp. from chevon and beef carcasses.
CONCLUSION
Even if, the microbial load of home slaughtered meat were low, dressing should be done and/or trends of slaughtering the meat should be adapted and done at abattoirs. The methods of slaughtering and storage conditions were the main reason for the processed meat contamination. These make the meat infected by pathogenic bacteria. To make the meat free of pathogenic bacteria the cattle should be dressed at abattoir and the standard operating methods should be practiced. Such methods include screening of operators, meat carriers and all who handle meat on regular basis on their health status. In addition well maintained meat rope, Garment, Garment rack, Hanging rack covered with nets, thoroughly cleaned and regularly sterilized knives and all equipments meats come into contact with should be used. Jimma (Ethiopia) is a tropical country, with ambient temperatures conducive for the growth of microorganisms, which can rapidly render meat unsafe for human consumption. So, appropriate slaughtering and storage materials were necessary for reduction of microorganism after dressing.
ACKNOWLEDGMENTS
I would like to express my deepest appreciation and special thanks to Ebba Alemayehu Biology Department head, Jimma University and Natural Science College for his, material and moral support to the accomplishment of this research work. My sincere appreciation and thanks goes to Shiferaw Demissie from Biology Department who stands behind me and provide unlimited personal support and professional assistance throughout.
REFERENCES
- Ali, N.H., A. Farooqui, A. Khan, A.Y. Khan and S.U. Kazmi, 2010. Microbial contamination of raw meat and its environment in retail shops in Karachi, Pakistan. J. Infect. Dev. Countries, 4: 382-388.
PubMedDirect Link - Dabassa, A. and K. Bacha, 2012. The prevalence and antibiogram of salmonella and shigella isolated from abattoir, Jimma Town, South West Ethiopia. Int. J. Pharm. Biol. Res., 3: 143-148.
Direct Link - Bacha, K., T. Mehari and M. Ashenafi, 2007. Microbiological study of wakalim, a traditional ethiopian fermented sausage. Ethiop. J. Biol. Sci., 6: 129-145.
Direct Link - Callaway, T.R., T.S. Edrington, R.C. Anderson, J.A. Byrd and D.J. Nisbet, 2008. Gastrointestinal microbial ecology and the safety of our food supply as related to Salmonella. J. Anim. Sci., 86: E163-E172.
CrossRef - Adzitey, F., G.A. Teye, A.G. Ayim and S. Adday, 2010. Microbial quality of chevon and mutton sold in Tamale Metropolis of Northern Ghana. J. Applied Sci. Environ. Manage., 14: 53-55.
Direct Link - Gruenewald, R., D.P. Dixon, M. Brun, S. Yappow and R. Henderson et al., 1990. Identification of salmonella somatic and flagellar antigens by modified serological methods. Applied Environ. Microbiol., 56: 24-30.
PubMedDirect Link - Iroha, I.R., E.C. Ugbo, D.C. Ilang, A.E. Oji and T.E. Ayogu, 2011. Bacterial contamination of raw meat sold in Abakaliki, Ebonyi State Nigeria. J. Public Health. Epidemiol., 3: 49-53.
Direct Link - Bryant, J., D.A. Brereton and C.O. Gill, 2003. Implementation of a validated HACCP system for the control of microbiological contamination of pig carcasses at a small abattoir. Can. Vet. J., 44: 51-55.
Direct Link - Bekele, J. and B. Butako, 2011. Occurrence and financial loss assessment of cystic echinococcosis (hydatidosis) in cattle slaughtered at Wolayita Sodo municipal abattoir, Southern Ethiopia. Trop. Anim. Health Prod., 43: 221-228.
CrossRefDirect Link - Kitcheil, A.G., 1962. Micnococci and coagulase negative Staphylococci in cured meats and meat products. J. Applied Microbiol., 25: 4l6-431.
CrossRefDirect Link - Mukhopadhyay, H.K., R.M. Pillai, U.K. Pal and V.J. Ajaykumar, 2009. Microbial quality of fresh Chevon and beef in retail outlets of Pondicherry. Tamilnadu J. Vet. Anim. Sci., 5: 33-36.
Direct Link - Nolan, C.M.E, L. Laborder, T.R. Howell and J.B. Robbins, 1980. Identification of Salmonella typhi in faecal specimens by an antiserum-agar method. J. Med. Microbiol., 13: 373-377.
PubMedDirect Link - Ogiehor, I.S., M.J. Ikenebomeh and A.O. Ekundayo, 2007. The bioload and aflatoxin content of market garri from some selected states in southern Nigeria: Public health significance. Afr. Health Sci., 7: 223-227.
Direct Link - Raji, A.I., 2006. Bacteriological quality of dried sliced beef (Kilishi) sold in Ilorin Metropolis. J. Applied Sci. Environ. Mgt., 10: 97-100.
Direct Link - Selvan, P., R.N. Babu, S. Sureshkumar and V. Venkataramanujam, 2007. Microbial quality of retail meat products available in Chennai city. Am. J. Food Technol., 2: 55-59.
CrossRefDirect Link - Ukut, I.O.E., I.O. Okonko, I.S. Ikpoh, A.O. Nkang and A.O. Udeze et al., 2010. Assessment of bacteriological quality of fresh meats sold in calabar metropolis, Nigeria. Electron. J. Environ. Agric. Food Chem., 9: 89-100.
Direct Link - Zakpaa, H.D., C.M. Imbeah and E.E. Mak-Mensah, 2009. Microbial characterization of fermented meat products on some selected markets in the Kumasi metropolis, Ghana. Afr. J. Food Sci., 3: 340-346.
Direct Link








