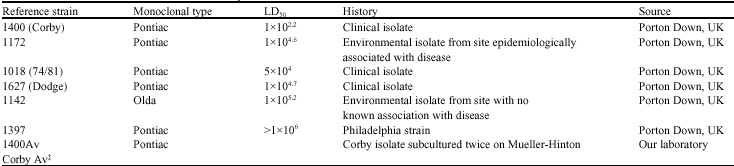
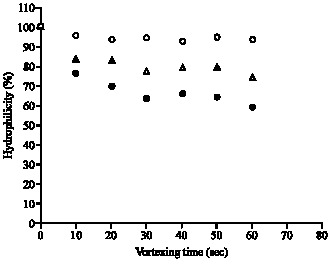
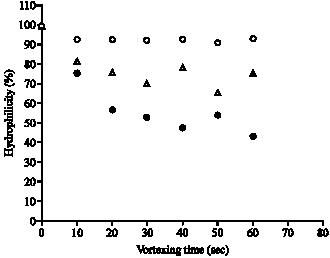
Research Article
Adherence of Virulent and Avirulent Legionella to Hydrocarbons
Department of Biological and Environmental Sciences, Faculty of Science, Beirut Arab University, Beirut, Lebanon
A. Al-Dahlawi
King Abdul Aziz University, Jeddah, Saudi Arabia