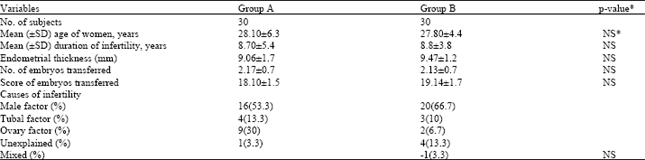
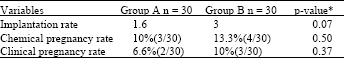Research Article
Transfer of Cryopreserved-Thawed Embryos in a Cycle Using Exogenous Steroids with or Without Prior Gonadotropihin-Releasing Hormone Agonist
Bouali Ave, Safaeyeh, Research and Clinical Center for Infertility, Yazd, Iran
Maryam Eftekhar
Clinical and Research Center for Infertility, Shahid Sacloughi University, YazcL Iran
Naeimeh Tayebi
Clinical and Research Center for Infertility, Shahid Sacloughi University, YazcL Iran