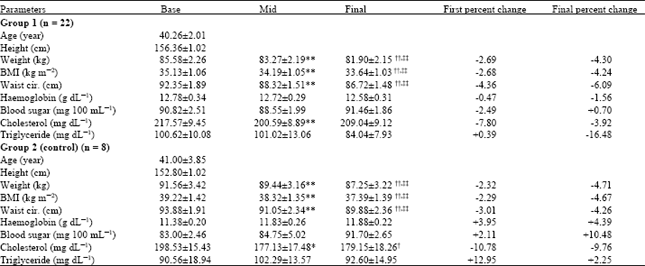
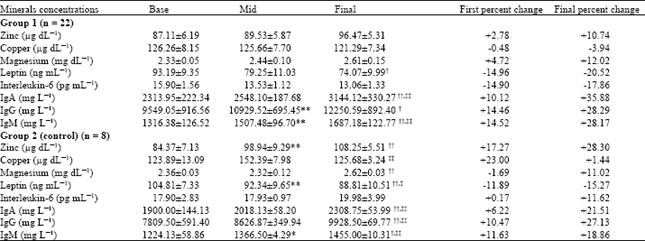
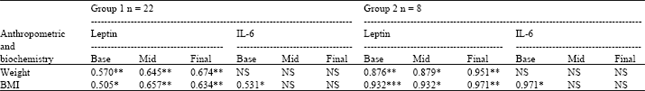
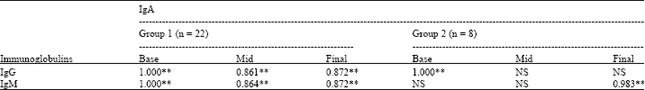
Research Article
Effects of High Vegetables Consumption on Inflammatory and Immune Functions in Human Obesity
Department of Food Science and Nutrition, National Research Center, Asian Network for Scientific Information Dokki, Cairo, Egypt
Salwa T. Tapozada
Department of Food Science and Nutrition, National Research Center, Asian Network for Scientific Information Dokki, Cairo, Egypt
Laila M. Hanna
Department of Food Science and Nutrition, National Research Center, Asian Network for Scientific Information Dokki, Cairo, Egypt
Lobna A. Ghattas
Department of Food Science and Nutrition, National Research Center, Asian Network for Scientific Information Dokki, Cairo, Egypt
Hamed I. Mohamed
Department of Food Science and Nutrition, National Research Center, Asian Network for Scientific Information Dokki, Cairo, Egypt