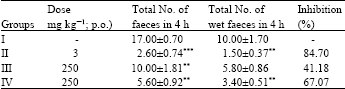
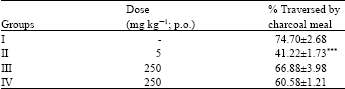
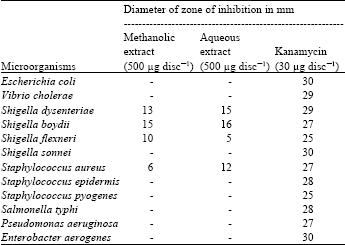
Research Article
Evaluation of Anti-diarrhoeal Activity of Enhydra fluctuans
Phytochemistry and Pharmacology Research Laboratory, Pharmacy Discipline, Life Science School, Khulna University, Khulna 9208, Bangladesh
M.M. Ferdous
Department of Botany, University of Rajshahi, Raj shahi 6205, Bangladesh Rhu]na Medical College, Khulna 9000, Bangladesh
R. Rouf
Phytochemistry and Pharmacology Research Laboratory, Pharmacy Discipline, Life Science School, Khulna University, Khulna 9208, Bangladesh
M.S. Alam
Phytochemistry and Pharmacology Research Laboratory, Pharmacy Discipline, Life Science School, Khulna University, Khulna 9208: Bangladesh
M.A.M. Sarkar
Department of Pharmacology and Therapeutics, Rhu]na Medical College, Khulna 9000, Bangladesh
J.A. Shilpi
Phytochemistry and Pharmacology Research Laboratory, Pharmacy Discipline, Life Science School, Khulna University, Khulna 9208, Bangladesh