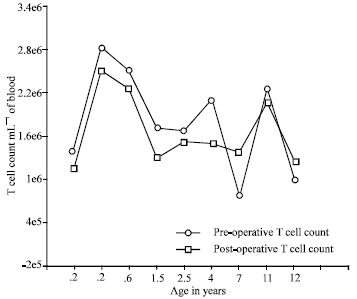
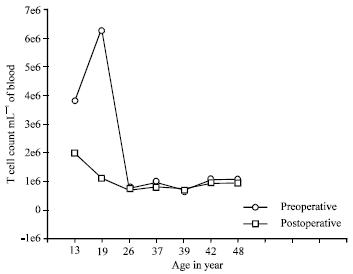
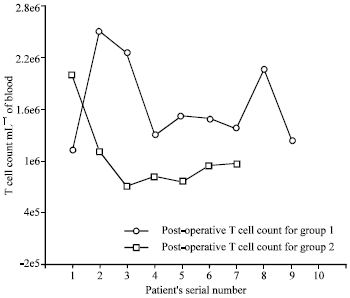
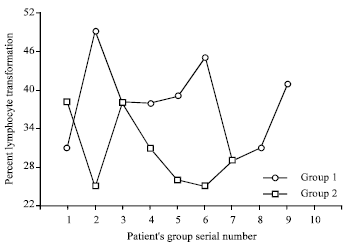
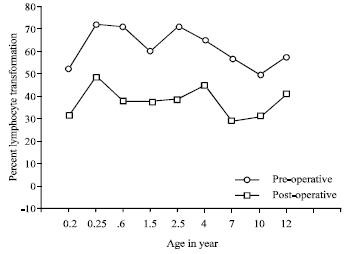
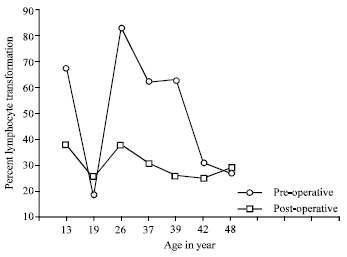
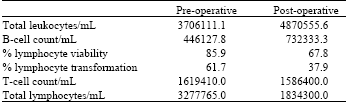
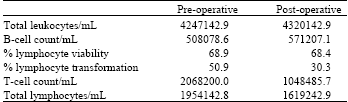
Research Article
The Influence of Age-Sex on the Pattern of Immunodepression Following Surgery
Department of Microbiology, Obafemi Awolowo University, Ile-lfe, Nigeria
O. Adejuyigbe
Department of Surgery, Obafemi Awolowo University, Ile-lfe, Nigeria
O.O. Shonukan
Department of Microbiology, Obafemi Awolowo University, Ile-lfe, Nigeria