Research Article
Discovery of the α-1 Microglobulin Complex in Urine Sample Patients with Cadmium Intoxication
Department of Biochemistry Faculty of Medicine Hasanuddin University Makassar, Indonesia
Plasma proteins with a molecular weight of <50 kDa are present only in quite low concentrations in blood of healthy individuals. Such proteins seem to pass the glomerular basement membrane in the kidney with relative easy and are probably reabsorbed and catabolized in renal tubules[1-6]. Only a small amount of plasma protein is normally excreted in final urine[3,6].
Alpha 1 microglobulin (α-1 m, also designated protein HC; human complex-forming glycoprotein, heterogeneous in charge) is a plasma glycoprotein carrying a brown-coloured chromophore and it was discovered since about fifty years ago in human urine[1-3] and is present in human plasma and other body fluids. It was first originally isolated from the urine of the patients of tubular proteinuria caused of chronic cadmium poisoining[1-4,7].
The molecular weight of α-1 m has been reported 25-30 kDa by sodium dodecyl sulfate polyacrylamide gel electrophoresis and migrated at the α-1 region on agarose gel electrophoresis[6] and it contains about 20% carbohydrate in their amino acid composition. Patients with tubular proteinuria due to renal tubular resorption defects excrete considerable amounts of low molecular weight plasma proteins[1-4,8-12].
Alpha 1 m is synthesized in the liver and after its secretion from the liver, approximately 50% of the α-1 m molecules are linked to other plasmaproteins including IgA, however on excretion into the urine, it is not found complexed with IgA[1-3,8]..
On the basis of the amino acid sequence, α-1 m belongs to the lipocalin superfamily, a group of extracellular proteins carrying small hydrophobic prosthetic groups. The super family also counts among its members retinal-binding protein, β-lactoglobulin, α-1 acid glucoprotein and odorant-binding nasal epithelial proteins[1-4,13].
Free α-1 m is filtrated through the glomerular membranes in the kidney and is then reabsorbed by tubular cells. Renal tubular disorders are easily diagnosed by elevated amounts of α-1 m in the urine[6,11].
The elevated levels of urinary α-1 m and β-2 m plasma from patients of renal tubular disfunction among lead workers have been reported[7].
In the human urine there are two forms of α-1 m but none of the α-1 m complexes are found in the urine[8].
The present study describes a low molecular weight α-1 m isolated from the urine patients with chronic cadmium intoxication, a condition known to be associated with tubular proteinuria and it showed a single band on SDS-PAGE. But this our result also contain a high molecular weight protein, 50-60 kDa showed by using Western-Blot analysis and this apparently novel glycoprotein is as called as α-1 microglobulin complex.
Urine: Twenty-four hours urine specimens were collected from eight patients with tubular proteinuria caused by chronic cadmium intoxication and one normal urine sample used as a control. All the patients from Toyama Perfecture Hospital, Toyama, Japan. All the urine sample patients and normal urine sample were ultrafiltrated and then concentrated to ten folds using Diaflo Ultrafiltration membranes (MA 01915 Amicon-USA, cut off point 10,000 MW).
Isolation of α-1 microglobulin
Gel chromatography: After ultrafiltrated, subsequently the urine sample were concentrated using ammonium sulfate between 20 and 80%. The concentrated urine of 5 mL was applied to the column chromatography on Sephadex G-100. The procedures were performed by the method described by EkstrAm[4].
Purification of α-1 microglobulin: Alpha 1 m were purified by affinity chromatography using CNBr-activated Sepharose-4B according to instructions provided by manufacturer (Pharmacia Biotech, Sweden). Briefly, 1.0 g freeze-dried Sepharose powder was suspended with 1 mM HCl and applied to the 10 mL tuberculin syringe used as a column. After washed with 1 mM HCl, rabbit IgG to α-1 m (Dako. Denmark) was added to the column chromato-graphy as a ligand by using coupling buffer, 0.1 M NaHCO3 pH 8.3 containing 0.5 M NaCl.
Then, washed away excess ligand with coupling buffer and then blocked the coupling. Transferred the gel to 0.1 M Tris-HCl buffer pH 8.0 and washed with 0.1 M Acetate buffer pH 4.0 containing 0.5 M NaCl followed by washed with 0.1 M Tris-buffer pH 8.0 containing 0.5 M NaCl and PBS pH 7.0. Sample urine of 2.5 mL was filtered then applied to the column. After the sample has been loaded, the elution of bound substances was done with 3 cycles buffer. PBS pH 7.0; Acetate buffer pH 4.0 and Glycine buffer pH 2.2. The concentration of the protein in the effluent was measured by reading the absorbance at 280 nm. Concentrated of α-1 m was measured by using fluorescence ELISA (FELISA). By affinity chromatography, the α-1 m used as a ligand and the highest concentrations were presumed as α-1 m.
SDS-polyacrylamide gel electrophoresis: SDS-polyacrylamide gel Electrophoresis were performed in 10-20% linear gradient gels by carried out the following method of Weber and Osborn[15] for estimation of molecular weight.
Immunoblotting analysis: Alpha 1 m bindings protein obtained from affinity chromatography were shown by SDS-PAGE and immediately transferred to polyvinylene difluoride (PVDF) membranes (Alto Corp, Japan). The membranes were incubated with rabbit-anti α-1 m antibody followed by alkaline phosphatase conjugated anti-rabbit IgG antibody and then staining with BCIP/NBT.
Purification of α-1 microglobulin: Alpha 1 m was isolated separately from several concentrated urine of the patients with chronic cadmium poisoning, which gave us similar results. Gel chromatography on sephadex G-100; 5 mL of concentrated urine (100 folded concentrated urine) were separated on column chromatography of sephadex G-100 (column 33x2 cm) equilibrated with 0.02 M Tris-HCl Buffer pH 7.8 containing 1.0 M NaCl. The fractions were collected at a flow rate of 24 mL h-1. The distribution of protein in the effluent was determined by reading the absorbancy at 280 nm. The peak protein were observed. The α-1 m was present in a highest protein peak (Fig. 1).
Fractions containing α-1 m were pooled and concentrated by ultrafiltration affinity chromatography on CNBr-activated Sepharose-4 B. The pooled α-1 m fractions from the gel column chromatography was dialyzed and applied to a CNBr-activated Sepharose-4B column. The purified α-1 m had a brown colour which could be observed during the entire isolation procedure. Purified α-1 m was shown on SDS-PAGE at about 30 kDa (Fig. 2).
SDS-polyacrylamide gel electrophoresis: Twenty four hours urine samples from eight patients with cadmium intoxication were collected and each urine sample was filtered and then dialyzed by used Mol-Cut off 10 kDa MW.
By SDS-PAGE with 10-20% gradient gel (Easy-Gel II), the molecular weight of each urine sample were performed at about 30 kDa and one other gel were immediately transferred to the polyvinyl difluoride/PVDF-membrane for immunoblotting.
This immunoblotting showed the high molecular weight of band at about 50-60 kDa of each line and no band was performed in the line of urine normal sample.
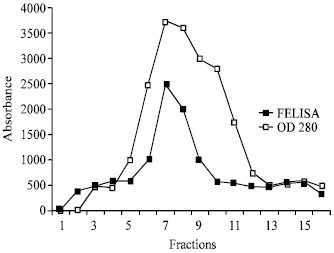 | |
| Fig. 1: | Elution pattern of urine sample from cadmium intoxication patients FELISA: Alpha 1 m concentration OD 28: Protein concentration |
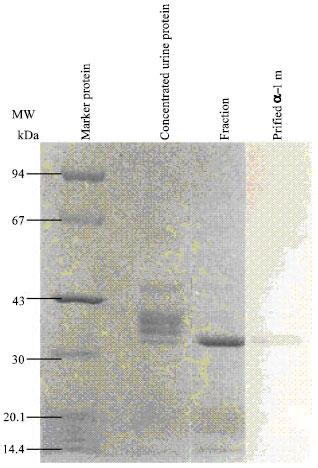 | |
| Fig. 2: | SDS-PAGE of purified urine patients Urine protein concentration by used ammonium sulfate between 20 and 80%. Fraction obtained by using Sephadex G-100 column chromatography and α-1 m purified by used affinity chromatography |
These results suggested that the α-1 m from the patient with cadmium intoxication were linked to other protein formed complexes (Fig. 3).
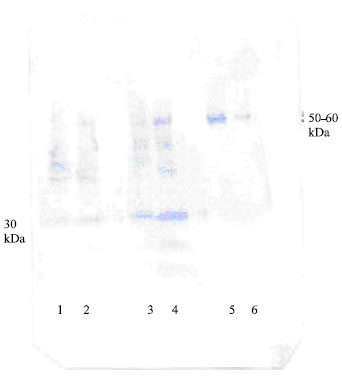 | |
| Fig. 3: | Western-immunoblot procedure of SDS-PAGE of crude urine samples reacted with rabbit antisera against α-1 m and developed with anti rabbit-lgG Alkaline-phosphatase. Free α-1 m with 30 kDa was showed in the lane 1-4 of gel chromatography fractions from crude urine patients. Lane 5-6 indicated that α-1 m complex with 50-60 kDa were showed and band of free α-1 m was not appeared |
The excretion of low molecular weight proteins or renal specific enzymes in urine may be a sensitive indicator of early renal injury. Alpha 1 m has been subsequently detected in normal human serum; normal urine and cerebrospinal fluid; and its serum concentration has been reported to increase in patients with renal insufficiency[4,6].
Urinary α-1 m and β-2 microglobulin have been measured among lead workers, but no one has reported the relationship between α-1 m and lead exposure[7]. By used immunochemical, both serum and urine from patients with renal disease had greatly increased concentrations of α-1 m and indicated that the kidney is its main catabolic sites[4,6].
The present communication reports on the purification of urinary α-1 m from patients with marked tubular proteinuria caused by cadmium intoxication and showed the peak protein in Fig. 1 by reading the absorbancy at 280 nm.
The lyophilized protein had a brown colour during the entire isolation procedure. The molecular weight of urinary α-1 m has been reported 25,000-30,000 Dalton and its contain about 20% carbohydrate[2,3,7-9,12-14]. This carbohydrate have been analyzed[4,13] and revealed close to equimolar amounts of glucosamine; mannose; galactose and sialic acid. In Fig. 2, by SDS-polyacrylamide showed one clear band of purified urinary α-1 m with about 30,000 molecular weight.
Alpha 1 m is also called protein HC (Heterogenous in charge) and showed charge heterogeneity by agarose gel electrophoresis or isoelectric focusing[6] and the complete amino acid sequence of α-1 m have been reported but have no found any amino acid sequence heterogeneity[1,2,4,9,11,16]. Alpha 1 m is present in human plasma as a free molecule as well as complexed to immunoglobulin A (IgA) in man or to α-1 macroglobulin inhibitor-3, a proteinase inhibitor of the macroglobulin superfamily in rat[8,14].
However, α-1 m belongs to so-called Lipocalin superfamily, a function as a lipophilic ligand carrier and that proteins are able to carry small lipophilic molecules such as retinol; odorants and steroids[1-3,8,12,14]. Also the lipocalins are predominantly extracellular transport protein which show high binding affinity[4,8,12,14].
Figure 3 suggested the α-1 m was complexed with other protein (MW. 50-60 kDa) that similarly showed to albumin. This high molecular weight complexed to α-1 m was detected by immunoblotting procedure used anti-rabbit α-1 m to human antibody.
Human urinary α-1 m (MW. 30 kDa) was purified and α-1 m complex was discovered. The molecular weight of human α-1 m complex was determined to about 50-60 kDa. However, we have not clearly identified yet, but the brown-colour material was shown during the entired isolation procedure. Present result suggested, the chromophore material seems to be quite strongly covalently binds to protein moiety as it was not removed by the various isolation procedure or by gel chromatography.
Due to relatively low molecular weight, it is filtrated through the renal glomeruli with larger amounts in urine. It is perhaps the most accurate marker of tubular proteinuria.
Biological role for the plasma protein α-1 m has yet to be defined, however, the true function of α-1 m is still unknown. Structurally, it belongs to the hydrophobic carrier protein superfamily lipocalins and forms covalently complexes with several other plasma proteins.
Hopefully, the further experiment are important for characterize the α-1 m complex and to identify the true function of both α-1 m and its complex.