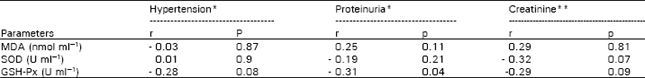
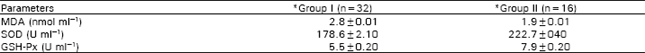Research Article
Oxidative Status in Children with Post-streptococcal Acute Glomrulonephritis
Department of Pediatrics, Mansoura University Children�s Hospital, Mansoura, Egypt
Ali Shaltout
Department of Pediatrics, Mansoura University Children�s Hospital, Mansoura, Egypt
Ayman Hammad
Department of Pediatrics, Mansoura University Children�s Hospital, Mansoura, Egypt
Amr Sarhan
Department of Pediatrics, Mansoura University Children�s Hospital, Mansoura, Egypt
Mohamed Abd El-Latef
Department of Clinical Biochemistry, Mansoura University Children�s Hospital, Mansoura, Egypt