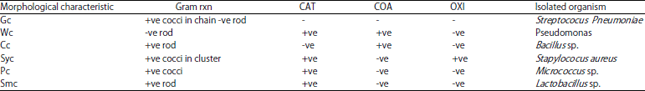
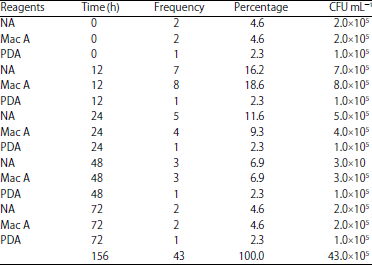
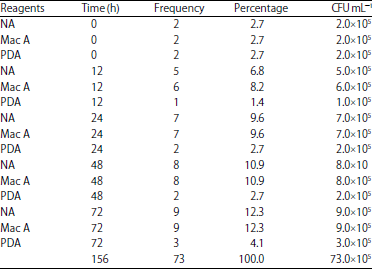
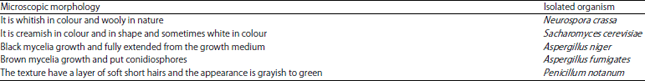
Research Article
Effect of Bitter Leaf Extract (Vernonia amygdalina) on Culturable Microorganisms Isolated from Palm Wine in Makurdi Metropolis
Department of Biological Sciences, University of Agriculture, Makurdi, Nigeria
T. Ichor
Department of Biological Sciences, University of Agriculture, Makurdi, Nigeria
Enyi Theresa Omeche
Department of Biological Sciences, University of Agriculture, Makurdi, Nigeria