Research Article
Endophytic Fungal Diversity in Tropical and Subtropical Plants
Department of Microbiology, Vidyasagar University, Midnapore, 721 102, West Bengal, India
Tropical and subtropical rainforests are found nearer to north or south of the equator. They are common in Asia, Australia, Africa, South America, Central America, Mexico and on many of the Pacific Islands (Fig. 1). Within the World Wildlife Fund's biome classification, tropical rainforests are considered a type of tropical wet forest and may also be referred to as lowland equatorial evergreen rainforest. Minimum normal annual rainfall between 1,750 mm (69 in) and 2,000 mL (79 in) occurs in this climate region. Mean monthly temperatures exceed 18°C (64°F) during all months of the year. Rainforests are home to half of all the living animal and plant species on the planet. Tropical rain forests are called the world's largest pharmacy because over one-quarter of modern medicines originate from its plants.
Tropical and temperate rainforests are the most biologically diverse terrestrial ecosystems on earth. The most threatened of these spots cover only 1.44% of the land’s surface, yet they harbor more than 60% of the world’s terrestrial biodiversity (Mittermeier et al., 1999). It is expected that areas of high plant endemicity also possess specific endophytes that may have evolved with the endemic plant species (Strobel, 2002a, b). Tropical rainforests are most active in evolutionary race to survive. Competition is great, resources are limited and selection pressure is at its peak. This gives rise to a high probability that rainforests are a source of novel molecular structures and biologically active compounds (Redell and Gordon, 2000). Bills et al. (2002) described a metabolic distinction between tropical and temperate endophytes through statistical data which compares the number of bioactive natural products isolated from endophytes of tropical regions to the number of those isolated from endophytes of temperate origin.
The most common usage of the term endophyte for organisms whose infections are internal and inconspicuous and in which the infected host tissues are at least transiently symptomless, is equally applicable to bacterial prokaryotes and fungal eukaryotes. Taken literally, the word endophyte means in the plant (endon = within, phyton = plant). Endophytes include an assemblage of microorganisms with different life history strategies: those that, following an endophytic growth phase, grow saprophytically on dead or senescing tissue, avirulent microorganisms, incidentals, but also latent pathogens and virulent pathogens at early stages of infection (Bacon and White, 2000; Suriyanarayanan et al., 2009). Communities of endophytes inhabiting a particular host may be ubiquitous, or have what is frequently referred to as host specificity (Carroll, 1988; Petrini, 1996; Stone et al., 2000).
 | |
| Fig. 1: | Tropical and subtropical rain forest (black areas in the map) |
Since, the number of plant species in the world is so great, creative and imaginative strategies must be used to quickly narrow the search for endophytes displaying bioactivity (Mittermeier et al., 1999). A specific rationale for the collection of each plant for endophyte isolation and natural-product discovery was proposed by Strobel and Daisy (2003). Hypotheses govern this plant selection strategy are as follows: (1) Plants from unique environmental settings, especially those with an unusual biology and possessing novel strategies for survival are seriously considered for study. (2) Plants that have an ethnobotanical history (use by indigenous peoples) that are related to the specific uses or applications of interest are selected for study. (3) Plants that are endemic, that have an unusual longevity, or that have occupied a certain ancient land mass, such as Gonwanaland, are also more likely to lodge endophytes with active natural products than other plants. (4) Plants growing in areas of great biodiversity also have the prospect of housing endophytes with great biodiversity. All of these selection strategies are applicable to plants of tropical and subtropical rain forests for endophyte isolation.
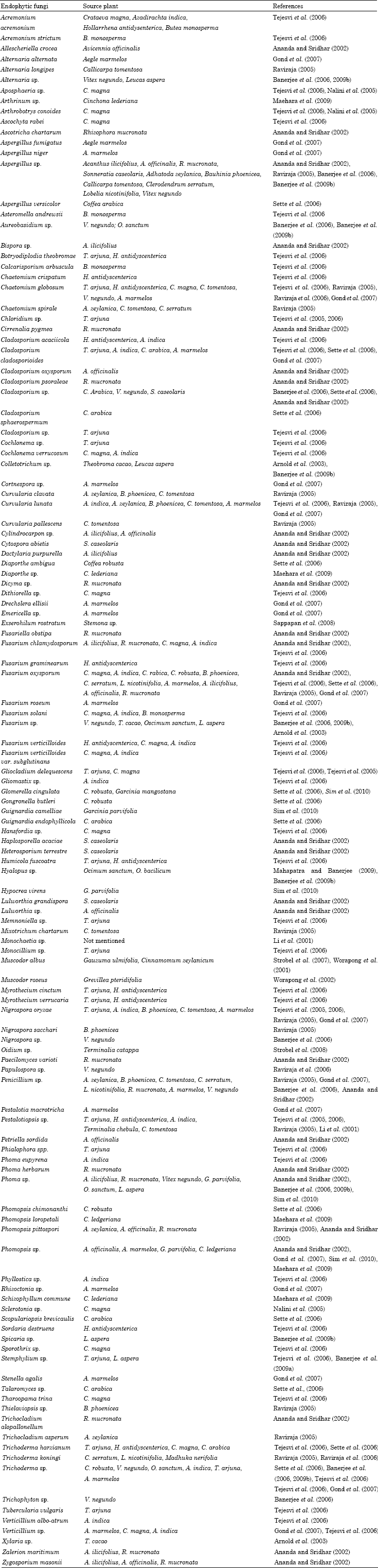
Tropical fungal endophytes: As tropical and subtropical climate harbor most of the worlds plants diversity so endophytic diversity in this climatic zone is also higher as almost all vascular plant species examined to date are found to posses endophytic bacteria and fungi (Firakova et al., 2007). The majority of undescribed fungal diversity lies within the tropical plant-associated fungi, yet the diversity and ecological role of endophytes in tropical angiosperms are almost entirely unexplored (Hawksworth, 1993; Rodrigues and Petrini, 1997). Endophytic fungal diversity has been studied essentially in the leaves, stems, petioles, barks and roots of many angiosperm taxa of the tropics (Table 1). Arnold et al. (2000) recovered 3000 fungal strains representing more than 418 morphospecies in the leaf tissue from two distantly related understory tree species of Heisteria concinna (Olacaceae) and Ouratea lucens (Ochnaceae) at Barro Colorado Island, Panama.
| Table 1: | Some fungal endophytes isolated from tropical and subtropical plants |
 | |
Reduced leaf damage and loss due to a major pathogen were reported in endophytic fungi associated woody angiosperm. Field surveys in lowland Panama to characterize the diversity, spatial structure and host affinity of natural endophyte infections in the economically important rainforest tree, Theobroma cacao (Malvaceae) were done. During this experiment Arnold et al. (2003) demonstrate that inoculation of leaf tissues by an assemblage of endophytes frequently isolated from naturally infected, asymptomatic hosts significantly reduces damage by an important foliar pathogen Phytophthora sp. Li et al. (2001) isolated Pestalotiopsis sp. and Monochaetia sp. from different plant materials collected in rainsforsts of several countries. They have isolated an antifungal agent, ambuic acid from those fungal endophytes. About 21 filamentous fungi were isolated from the young stems of a rubiaceaceous plant Cinchona ledgeriana in West Java, Indonesia (Maehara et al., 2009). The isolates belongs to six genera are Phomopsis sp., Diaporthe sp., Schizophyllum sp., Penicillium sp., Fomitopsis sp. and Arthrinium sp. About 24 endophytic fungi were isolated from Garcinia mangostana and Garcinia parvifolia plants in Sungai Rengit village, Johar, Malaysia. Isolated endophytes were characterized through internal transcribed spacer (ITS) region sequence analysis (Sim et al., 2010). Gond et al. (2007) reported 79 isolates belonging to 21 fungal genera from bark, leaf and root parts of Aegle marmelos in Baranasi, India. Most of the fungal isolates belong to hyphomycetes. They have reported a large number of Alternaria alternate and Fusarium roseum as fungal endophyte among the isolates. Different medicinal plants were screened to isolate fungal endophytes in Western Ghats of India (Raviraja, 2005; Raviraja et al., 2006). Curvularia and Fusarium were found in maximum number among 18 different endophytic fungal isolates. Tejesvi et al. (2006) studied the fungal diversity of some medicinal trees of southern India. They have isolated 48 fungal species from bark samples of Terminalia arjuna, Crataeva magna, Azadirachta indica, Holarrhena antidysenterica, Terminalia chebula and Butea monosperma. Ananda and Sridhar (2002) studied endophytic fungal diversity of mangrove species in west coast of India and reported 35 fungal species including 4 sterile ones. They have reported highest species richness in Rhizophora mucronata. Endophyte assemblage was also reported in another mangrove plant Rhizophora apiculata by Kumaresan and Suriyanarayanan (2002). Suryanarayanan and Rajagopal (2000) isolated 963 fungi from the bark sample of 10 tropical tree species in southern India. Banerjee et al. (2006) isolated different endophytic fungi from Vitex negundo in Midnapore, West Bengal, India. Banerjee et al. (2009a, b) reported presence of 14 endophytic fungal genera in three medicinal herb Ocimum sanctum, Ocimum bacilicum and Leucus aspera. Sette et al. (2006) isolated 39 strains of endophytic fungi from coffee plants in Pindorama, Sao Paulo, Brazil and identified the isolates using ITS region sequence analysis. Sixty-nine fungal species were isolated from the bark of a single Coprinus caroliniana tree, which suggest the enormous extent of fungal diversity in a single plant (Bills and Polishook, 1991). One of the potent volatile antibiotic producing fungi Muscodor albus was isolated from Cinnamomum zeylanicum in a botanical garden in Honduras (Strobel, 2006; Strobel et al., 2007). Other isolates of this fungus have been obtained in rainforests ranging from the upper Amazon countries, to Indonesia, Thailand and Australia (Mitchell et al., 2010; Banerjee et al., 2010).
Tropical and subtropical rainforest are most rich in plant diversity so endophytic diversity in this region is highest. Most of the work done till date with endophytic fungi is from the plants of this region. Rapid diminishment of plants from this highly diverse region needs special attention otherwise we will loose a great microbial resource in near future.
Author is thankful to University Grants Commission (UGC), New Delhi for financial assistance in major research project.