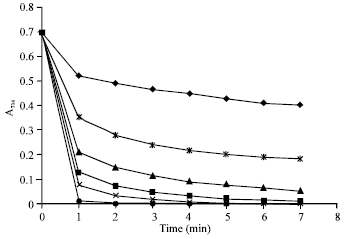
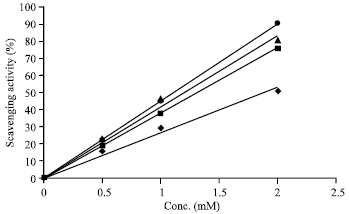
Research Article
Antioxidant Activity Improvement of Soybean Meal by Microbial Fermentation
Biotechnology Program, Graduate School, Chiang Mai University, Thailand
Chartchai Khanongnuch
Department of Biotechnology, Faculty of Agro-Industry, Chiang Mai University, Thailand
Puntipa Pongpiachan
Department of Animal Science, Faculty of Agriculture, Chiang Mai University, Thailand
Saisamorn Lumyong
Department of Biology, Faculty of Science, Chiang Mai University, Thailand