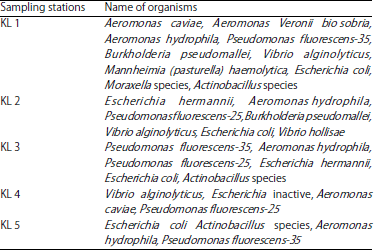
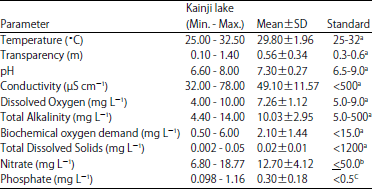
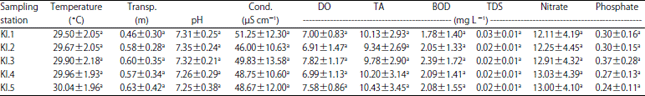
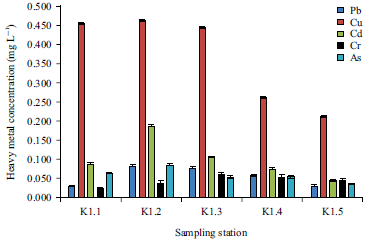
Research Article
Limnological Qualities and Heavy Metal Status of Water from Southern Basin of Kainji Lake, Nigeria
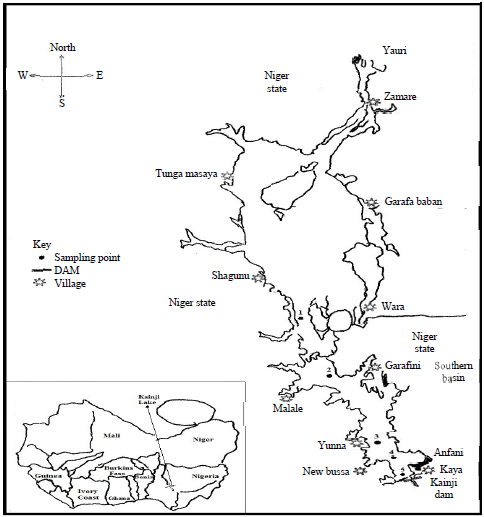
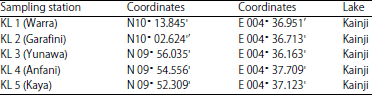
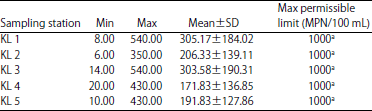
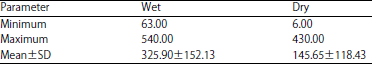
National Institute for Freshwater Fisheries Research, PMB 6006, New Bussa, Niger state, Nigeria
LiveDNA: 234.33367
Abioye O. Peter
Department of Microbiology, Federal University of Technology, PMB 65, Minna, Nigeria
Safiya Y. Daniyan
Department of Microbiology, Federal University of Technology, PMB 65, Minna, Nigeria
Ojutiku O. Rashed
Department of Fisheries, Federal University of Technology, PMB 65, Minna, Nigeria
Yem I. Yakubu
National Institute for Freshwater Fisheries Research, PMB 6006, New Bussa, Niger state, Nigeria