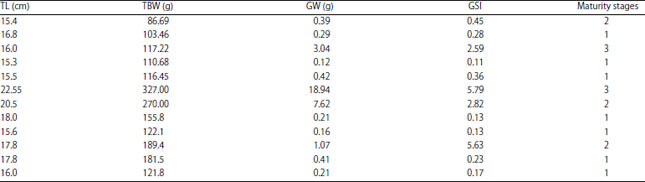
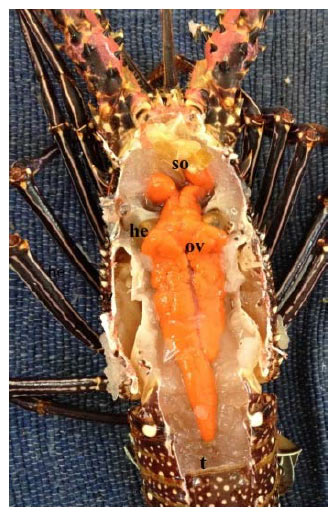
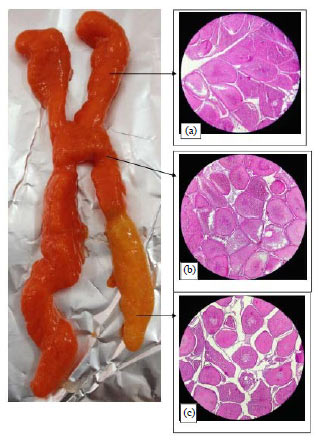
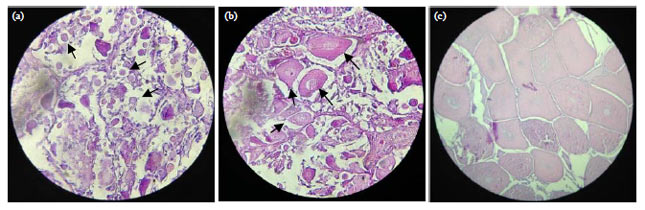
Research Article
Morphology of the Female Gonads of the Long-legged Spiny Lobster
Faculty of Fisheries and Marine Science, Halu Oleo University, 93232 Kendari, Indonesia
Indriyani Nur
Faculty of Fisheries and Marine Science, Halu Oleo University, 93232 Kendari, Indonesia
LiveDNA: 62.27287
Muhammad Idris
Faculty of Fisheries and Marine Science, Halu Oleo University, 93232 Kendari, Indonesia
Farid Yasidi
Faculty of Fisheries and Marine Science, Halu Oleo University, 93232 Kendari, Indonesia