ABSTRACT
The 2661 specimens of the striped mullet, Mugil cephalus L., used for this study, were collected from high brackish lagoon in south-west Nigeria for 24 months. The size of the 2661 specimens of M. cephalus used for this study ranged from 1.7-29.5 cm standard length (total length: 1.9-39.0 cm). The sex ratio for M. cephalus was 1:0.53 and this showed a statistically significant (p<0.05) dominance of the males over the females for the size range. The fecundity estimates varied from 635,568-1,520,185 and was positively correlated to the fish length and weight. Oocyte diameter averaged 409.64±40.67 μm and this is indicative of an early stage of gonad maturation of the specimens in the lagoon before final spawning occurred. The GSI indicated that spawning activity occurred from December to May in the open ocean.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jfas.2014.455.462
URL: https://scialert.net/abstract/?doi=jfas.2014.455.462
INTRODUCTION
Mullets (Mugilidae) are common fishes in the coastal waters of tropical and subtropical countries of the world. Sixteen species have so, far been identified in West Africa (Fowler, 1936; Cadenat, 1954; Blay, 1995) and these constitute an important proportion of the catches of commercial and subsistence fishermen in some countries in this area (Brulhet, 1975; Payne, 1976). General information on the biology of striped mullet has been well documented (Anderson, 1958; Thomson, 1963, 1966; Chubb et al., 1981) but limited information is available on the reproductive biology of wild populations (Anderson, 1958; Greeley et al., 1987; Render et al., 1995). Ditty and Shaw (1996) reported that Mugil cephalus are isochronal spawners, with all oocytes reaching maturity at the same time. It sheds its eggs in batches (Silva and de Silva, 1981; Greeley et al., 1987; Render et al., 1995). A thorough knowledge of the fecundity of fish is essential for evaluating the commercial potentialities, stock study, life history study, practical culture and actual management of the fishery (Das, 1977; Rheman et al., 2002). A few reports are available on the biology of mullet species in the coastal waters of West Africa and were principally on the food and feeding habits (Facade and Olaniyan, 1973; Brulhet, 1975; Blay, 1995; Soyinka, 2008). Information on the reproductive biology of mullet species is essential to understanding the population dynamics and the recruitment pattern of the fish, to harness it for conservation and management. The present investigation reports on the sex ratio, fecundity and the gonadosomatic index of M. cephalus from a high brackish lagoon in Nigeria, West Africa.
MATERIALS AND METHODS
Description of study area: The Lagos Lagoon (Fig. 1) which lies between longitudes 3° 20' and 3° 40'E and latitudes 6° 15' and 6° 40’ has an area of 208 km2 and is the largest of the lagoon systems of the West African sub-region.
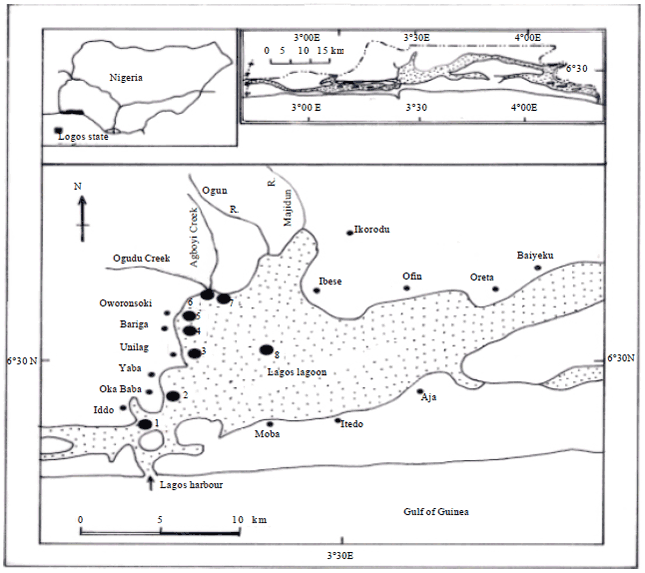 | |
| Fig. 1: | Map of Lagos Lagoon, Nigeria showing the sampling stations (1-8) |
It is the largest of the nine coastal lagoons of south-western Nigeria (others are Yewa, Badagry, Iyagbe, Ologe, Kuramo, Epe, Lekki and Mahin Lagoons) (FAO., 1969). It has supported decades of small scale fisheries which have shown sign of continuous decline (Solarin, 1998). The lagoon characterized with seasonal fluctuation in salinity-high brackish water during the dry season (December-May) while freshwater condition exist in the rainy season (June-November) (Facade and Olaniyan, 1974; Kusemiju, 1975; Solarin, 1998). The lagoon is surrounded by swamp forest and riparian forest consisting of mangrove vegetation: Rhizophora racemosa, R. harrisonii, Aerosticum aureum, Paspalum orbiculare, Langucularia sp. and Avicennia germinans. The lagoon empties into the Atlantic Ocean via the Lagos Harbour (Ogunwenmo and Kusemiju, 2004). The lagoon is shallow in depth and in most places a little more than 1.5 m depth (Solarin and Kusemiju, 2003).
Field studies: The specimens of the grey mullet, Mugil cephalus were collected from the Lagos Lagoon by fisher-folks who use active fishing gears for fishing. The collections were made fortnightly between February 2004 and January 2006. Fish were placed in ice-chest on the field but later transferred into a deep freezer at temperature of -20°C in the laboratory for further analysis. The salinity of the lagoon was determined in-situ twice a month between 8.00-11.00 a.m for 24 months using a Refractometer (BIOMARINE, Aqua Fauna Model).
Laboratory procedures: The specimens were removed from the freezer and allowed to thaw before being examined. Excess water from the fish was drained away or by using filter paper. The Standard Length (SL) and Total Length (TL) of the specimens were measured on a measuring board to the nearest 0.1 cm. The total weight of the fish was taken on a ‘Sartorious’ top loading balance (Model 1106 2608053) or a triple beam balance to the nearest tenth of a gram.
Sex determination: The fish were separated into males, females and juvenile or immature fish after dissecting the fish and observing the genital papillae visually. The male has a pair of thread-like tubes with whitish milt, the female has a pair of bulging or sac-like ovaries while the gonad of immature fish could not be differentiated with the naked eye.
Fecundity and egg diameter: Females with ripe ovaries were used for this study. The ripe ovaries were removed carefully from the abdominal region of the fish, the weight measured on the balance, before preserving in 10% formalin or 4% Gilson’s fluid. The eggs were separated from the ovarian tissue into petri dish. Two methods were used for the enumeration. By gravimetric method, the number of eggs in a known weight was used to estimate the total number in the dry sample. By volumetric method, the number of eggs in 1 mL using ‘Gilson Pipetman’ egg counting device (Model: D-82-13103) were counted under a microscope and used to estimate the total number in the remaining volume. The relationship between the fecundity, length and weight was expressed as:
Y = a+bX
Where:| Y | = | Fecundity estimate |
| X | = | Standard length (cm)/total weight (g) |
| a | = | Regression constant |
| b | = | Regression coefficient |
Egg diameter: For the egg diameter, eggs were placed on a slide, observed under a graduated eye-piece of a light microscope and the average diameter recorded.
Gonadosomatic index: This indicates gonadal development and maturity of fish. It increases with the maturation of fish declining abruptly thereafter (Rheman et al., 2002; Parameswarn et al., 1974; McDonough et al., 2003). This was estimated thus:
RESULTS
Sex ratio: The 2661 specimens of M. cephalus collected from the Lagos Lagoon were made up of 426 males and 224 female fish giving a sex ratio of 1: 0.53 (male/female). The calculated Chi-square (χ2) test showed that the males were significantly (p<0.05) more abundant than the females in Lagos Lagoon.
The monthly sex ratio values showed that in Lagos Lagoon (Fig. 2), the males were more than the females throughout the period of study except in the dry season of February 2004 and December 2004. They were nearly the same in the dry season of April 2004. Statistically, Chi-square (χ2) values of sex ratio were significantly different in February and July 2004, April, May, June, July, August, September and October 2005.
The relationship between the sex ratio and the salinity fluctuations which is the critical physico-chemical parameter affecting the distribution of species in the Lagos Lagoon is illustrated in Fig. 3. The result showed that immature specimens were predominant in the low brackish to fresh water condition of the lagoon; males were significantly more than females in most months while the population of females was more abundant during dry season than the wet season.
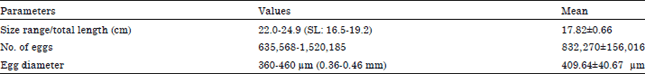
Fecundity of M. cephalus: The number of fecund females collected throughout the sampling period was very few. Only 28 fecund M. cephalus specimens from the Lagos Lagoon were examined for fecundity. They ranged from 16.5-19.2 cm standard length (total length, 22.0-24.9 cm) and weighed between 90.00-145.99 g. The fecundity estimates varied from 635,568-1,520,185 eggs.
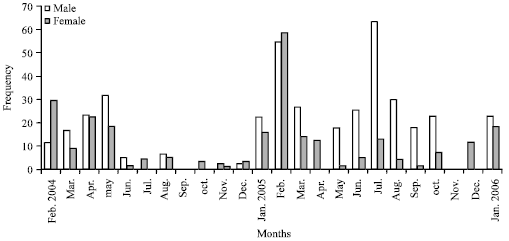 | |
| Fig. 2: | Monthly variation in sex ratio in M. cephalus from Lagos Lagoon (February 2004 to January 2006) |
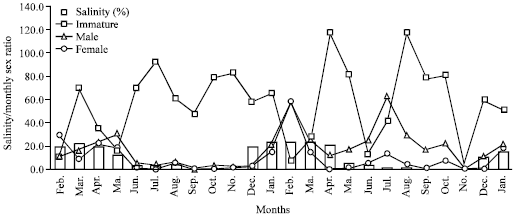 | |
| Fig. 3: | Sex ratio/salinity relationship for M. cephalus in Lagos Lagoon (February 2004 to January 2006) |
An average fecundity estimate was 832,270±156,016 eggs. The log fecundity-log standard length and log fecundity-log weight relationships are shown in Fig. 4 and 5, respectively.
A slightly high correlation existed between fecundity and the standard length (r = 0.5423) and the weight (r = 0.5054) of the fish.
The linear regression equations were:
| • | Log fecundity = 3.7751+1.7039 log standard length (n = 28, r = 0.5423) |
| • | Log fecundity = 4.8541+0.5168 log total weight (n = 28, r = 0.5054) |
Egg diameter of M. cephalus: The diameter of eggs of mature females in the Lagos Lagoon ranged from 360-460 μm (0.36-0.46 mm) as shown in Table 1. The least total size of female fish with ripe gonad was 22.0 cm (standard length: 16.5 cm).
| Table 1: | Diameter of eggs in mature females of M. cephalus from Lagos Lagoon |
 | |
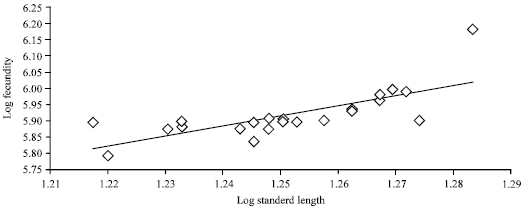 | |
| Fig. 4: | Log standard length/log fecundity relationship of M. cephalus from Lagos Lagoon |
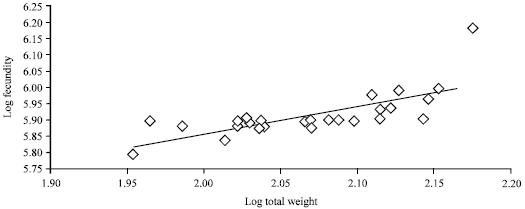 | |
| Fig. 5: | Log weight/log fecundity relationship of M. cephalus from Lagos Lagoon |
Gonadosomatic index: The Gonadosomatic Index (GSI) of 28 specimens of M. cephalus from the Lagos Lagoon was examined. The GSI values ranged from 6.45-10.00%. The mean value was 8.16±0.97. This indicated that M. cephalus on the average used 8.16% of its body weight for egg production.
DISCUSSION
In the sex ratio of M. cephalus collected from the Lagos Lagoon, the males were significantly more than the females as shown in the calculated Chi square (χ2) test. This indicated that probably, more male M. cephalus occur in the Lagos Lagoon than the females. McDonough et al. (2005) recorded a male to female ratio of 2:1 until the fish were larger than 32.5 cm TL. The ratio was 1:3.8 for fish greater than 32.5 cm TL. The ratio of 2:1 in South Carolina estuary almost agreed with the present study from the Lagos Lagoon. Mature females with ripening and ripe gonads were collected often in the dry season (December-May) with peak between late January and March, when salinity is high in the Lagos Lagoon. Facade and Olaniyan (1974) reported the availability of the fish during brackish (0.5-28.0%) condition of Lagos Lagoon. The monthly variation in sex ratio of the species from Lagos Lagoon showed that the males were more than the females except in February 2004 and December 2004 which fall within the dry season. In Fig. 3, there was an increase in number of female fish caught in the dry season (high salinity) than the wet season (low salinity). This coincided with the period females with ripe gonads were found in the collection. Male specimens, with sexually differentiated gonads also had a slightly similar seasonal distribution pattern but the immature ones were more available at both seasons. This depicted that salinity does influence gonad maturation in the grey mullet. McDonough et al. (2005) reported that striped mullet are sexually undifferentiated for the first 12 months, began differentiation at 13 months and are 90% fully differentiated by 15-19 months of age and 22.5 cm TL. The abundance of 1 and 2 years old striped mullet in South Carolina indicated that immature fish dominated the estuarine population. This agreed with the present study.
In this study, an average fecundity estimate of 832,270 eggs (~0.83 million eggs) was recorded for fish ranging in standard length from 22.0-24.9 cm and weight 90.00-145.99 g. Fecundity was averagely correlated with standard length and body weight. Silva and de Silva (1981) recorded a fecundity of 0.45-4.2 million in M. cephalus ranging in length from 32-56 cm and weight from 0.7-2.2 kg, fecundity was significantly correlated to body length, body weight and gonad weight. Collins (1985) estimated fecundity of striped mullet at 0.5-2.0 million eggs per female, depending on the size of the female. Ditty and Shaw (1996) reported that Mugil cephalus are isochronal spawners, with all oocytes reaching maturity at the same time. It sheds its eggs in batches (Silva and de Silva, 1981; Greeley et al., 1987; Render et al., 1995). The minimum spawning size of females is between 31 and 34 cm (Ditty and Shaw, 1996) while Silva and de Silva (1981) recorded female maturity at 31.5 cm and males at 34.0 cm. McDonough et al. (2005) recorded male spawning size at 24.8-30.0 cm while the spawning size for the female was 29.1-40.0 cm. This implied that the specimens in the present investigation were still at the early stage of gonad maturation and would not spawn possibly until they embark on the open ocean spawning migration where the gonads reach final maturation. Size at maturity has been found to range widely from 23.0 cm standard length (Thomson, 1963; Greeley et al., 1987) to 41.0 cm SL (Thomson, 1963; Chubb et al., 1981) for two and three-year-old fish. The slightly earlier lower end of the maturation size of fish recorded in the present study could probably be as a result of the tropical climate of the Lagos Lagoon.
The measurement of the diameter of the ripe eggs of mature females of M. cephalus from the Lagos Lagoon showed slight variation in egg sizes. McDonough et al. (2003) recorded oocyte diameter of 463-682 μm (mean size, 596 μm). Rottmann et al. (1991) reported an approximate mature egg diameter of 0.6-0.8 mm for Mugil cephalus. The slight variation of the lower end of the egg diameter in the present study is because of the size of fish used.
McDonough et al. (2003) reported a GSI value rang of 7.7-27.7 for fecund specimens in South Carolina estuary. The value of 8.16 recorded in this study falls within that range. It is clear that the gonad weight had a considerable influence on total body weight in fecund specimens (McDonough et al., 2003). The value of the GSI levels calculated in fish during the dry season (December-May) with peak between late January and March, showed that reproductive activity is within that period. McDonough et al. (2003) reported that striped mullet that recruit into South Carolina estuaries spawn from October through April. The information provided in the present study would be helpful in the adequate management of the M. cephalus stock in the Lagos Lagoon.
REFERENCES
- Blay, Jr. J., 1995. Food and feeding habits of four species of juvenile mullet (Mugilidae) in a tidal lagoon in Ghana. J. Fish Biol., 46: 134-141.
CrossRefDirect Link - Chubb, C.F., I.C. Potter, C.J. Grant, R.C.J. Lenanton and J. Wallace, 1981. Age, stucture, growth rates and movements of sea mullet, Mugil cephalus L. and yellow-eye mullet, Aldrichetta forsteri (Valenciennes), in the swan-avon river system, Western Australia. Aust. J. Mar. Freshwater Res., 32: 605-628.
Direct Link - Das, H.P., 1977. The fecundity of grey mullet Mugil cephalus L. along the Goya coast. Mahasagar Bull. Natl. Inst. Oceanogr., 10: 79-82.
Direct Link - Ditty, J.G. and R.F. Shaw, 1996. Spatial and temporal distribution of larval striped mullet (Mugil cephalus) and white mullet (M. curema , Family: Mugilidae) in the northern gulf of Mexico, with notes on mountain mullet, Agonostomus monticola. Bull. Mar. Sci., 59: 271-288.
Direct Link - Facade, S.O. and C.I.O. Olaniyan, 1973. The food and feeding interrelationship of the fishes in the Lagos Lagoon. J. Fish Biol., 5: 205-225.
CrossRefDirect Link - Greeley, Jr. M.S., D.R. Calder and R.A. Wallace, 1987. Oocyte growth and development in the striped mullet, Mugil cephalus, during seasonal ovarian recrudescence: Relationship to fecundity and size at maturity. Fish. Bull., 85: 187-200.
Direct Link - McDonough, C.J., W.A. Roumillat and C.A. Wenner, 2003. Fecundity and spawning season of striped mullet (Mugil cephalus L.) in South Carolina estuaries. Fish. Bull., 101: 822-834.
Direct Link - McDonough, C.J., W.A. Roumillat and C.A. Wenner, 2005. Sexual differentiation and gonad development in striped mullet (Mugil cephalus L.) from South Carolina estuaries. Fish. Bull., 103: 601-619.
Direct Link - Ogunwenmo, C.A. and K. Kusemiju, 2004. Annelids of a West African estuarine system. J. Environ. Biol., 25: 227-237.
Direct Link - Payne, A.I., 1976. The relative Abundance and feeding habits of the grey mullet species occurring in an estuary in Sierra Leone, West Africa. Mar. Biol., 35: 277-286.
CrossRefDirect Link - Render, J.H., B.A. Thompson and R.L. Allen, 1995. Reproductive development of striped mullet in Louisiana estuarine waters with notes on the applicability of reproductive assessment methods for isochronal species. Trans. Am. Fish. Soc., 124: 26-36.
CrossRef - Rheman, S., M.L. Islam, M.M.R. Shah, S. Mondal and M.J. Alam, 2002. Observation on the fecundity and gonadosomatic index (GSI) of Grey mullet Liza parsia (Ham.). J. Biol. Sci., 2: 690-693.
CrossRefDirect Link - Silva, E.I.L. and S.S. de Silva, 1981. Aspects of the biology of grey mullet, Mugil cephalus L., adult populations of a coastal lagoon in Sri Lanka. J. Fish Biol., 19: 1-10.
CrossRefDirect Link - Solarin, B.B. and K. Kusemiju, 2003. An appraisal of gender participation in trap and liftnet fisheries in Lagos Lagoon, Nigeria. Afr. J. Applied Zool. Environ. Biol., 5: 75-81.
Direct Link - Soyinka, O.O., 2008. The feeding ecology of Mugil cephalus (Linnaeus) from a high brackish tropical lagoon in South-west, Nigeria. Afr. J. Biotechnol., 7: 4192-4198.
Direct Link








