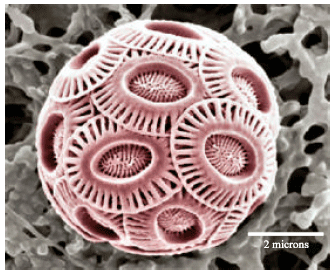
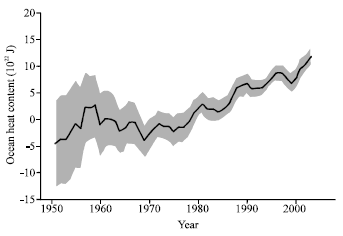
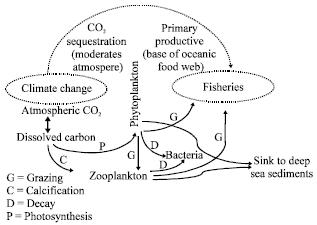
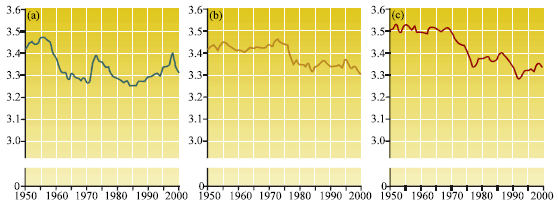
Review Article
Linking Climate Change and Fisheries: The Role of Phytoplankton
Department of Fisheries, Lagos State University, P.M.B 0001 LASU, Ojo, Lagos, Nigeria
M.A. Adewolu
Department of Fisheries, Lagos State University, P.M.B 0001 LASU, Ojo, Lagos, Nigeria
E.O. Lawson
Department of Fisheries, Lagos State University, P.M.B 0001 LASU, Ojo, Lagos, Nigeria