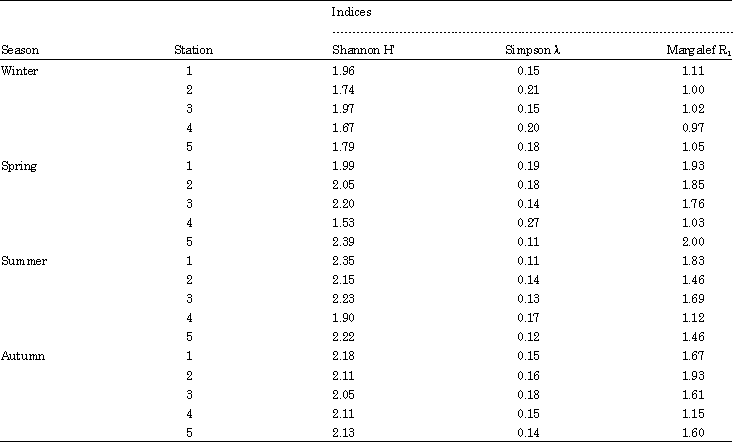
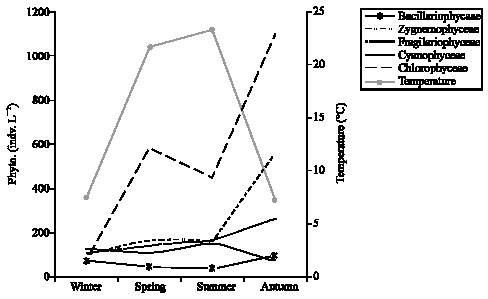
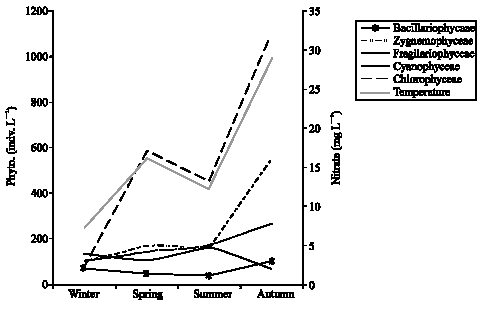
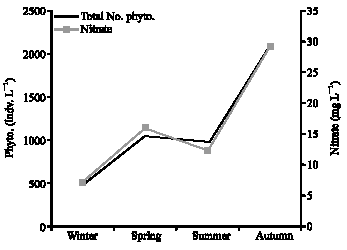
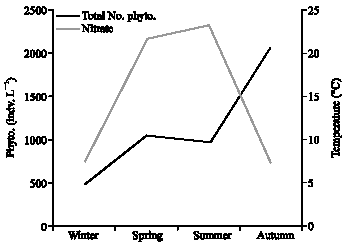
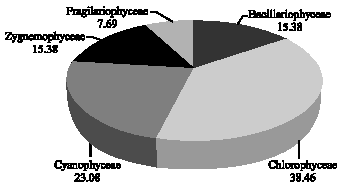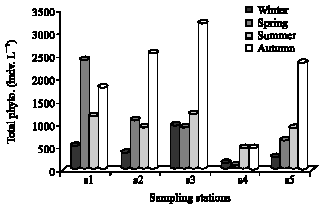Research Article
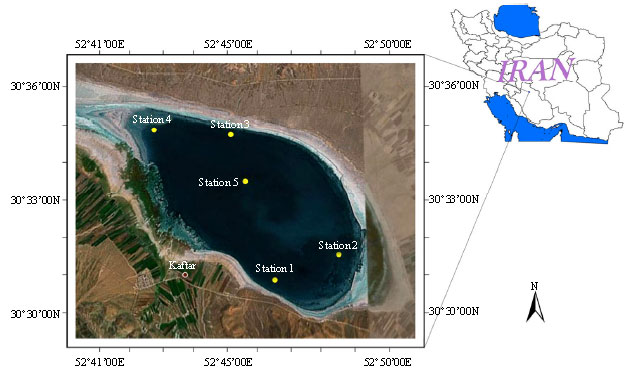
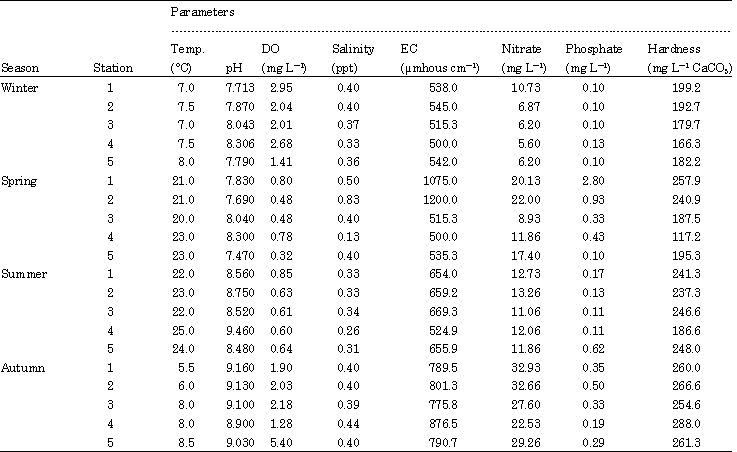
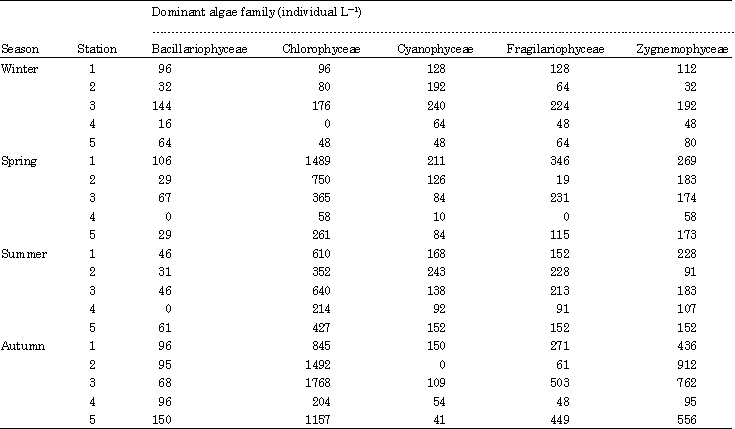
Effects of Environmental Factors on Phytoplankton Abundance and Diversity in Kaftar Lake
Department of the Environment, Shiraz, Fars, Postal Code 71346-53185, Iran
H. Valavi
Department of Marine Biology, Faculty of Marine Science, Khorramshahr University of Marine Science and Technology, P.O. Box 669, Khorramshahr, Khuzestan, Iran