Research Article
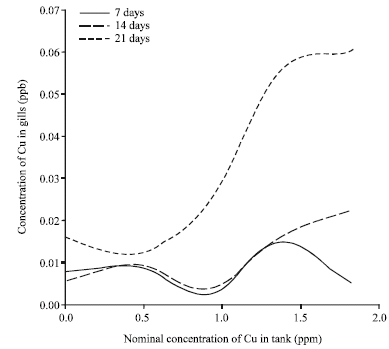
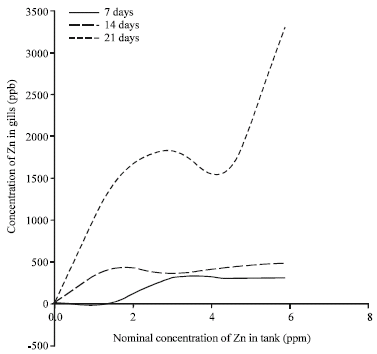
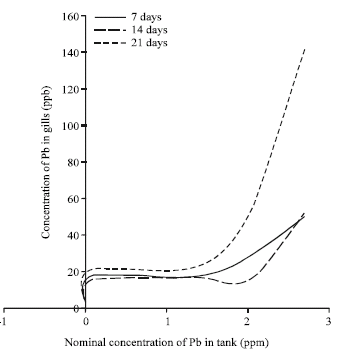
Accumulation of Metals in the Gills of Tilapia Fingerlings (Oreochromis niloticus) from in vitro Toxicology Study
International Islamic University Malaysia, Jalan Istana, Bandar Indera Mahkota, 25200, Kuantan, Pahang, Malaysia
B. Akbar
International Islamic University Malaysia, Jalan Istana, Bandar Indera Mahkota, 25200, Kuantan, Pahang, Malaysia
K.C.A. Jalal
International Islamic University Malaysia, Jalan Istana, Bandar Indera Mahkota, 25200, Kuantan, Pahang, Malaysia
S. Shahbudin
International Islamic University Malaysia, Jalan Istana, Bandar Indera Mahkota, 25200, Kuantan, Pahang, Malaysia