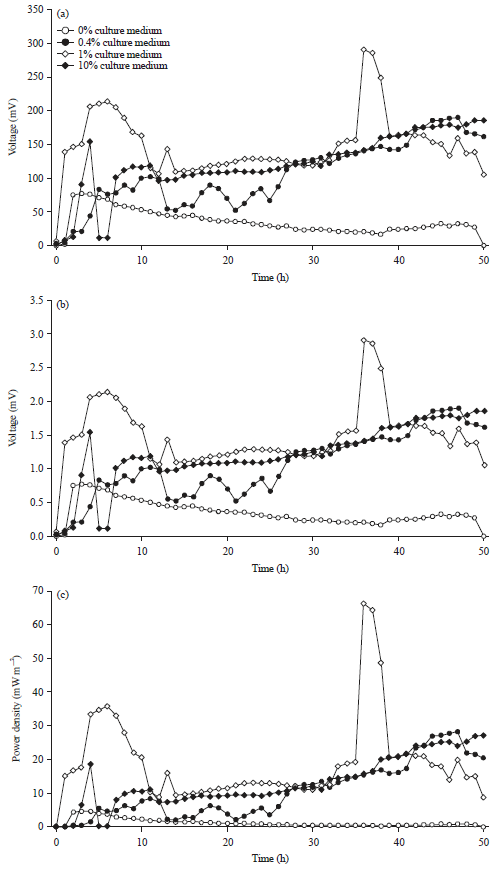
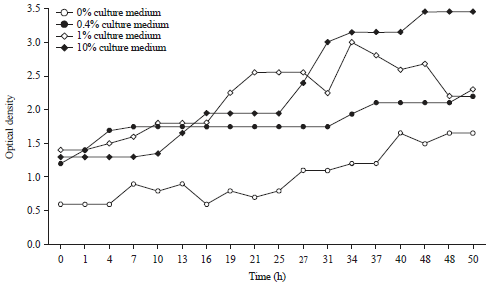
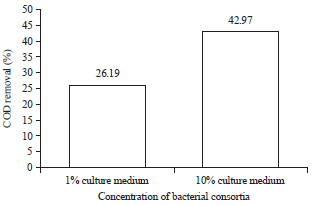
Research Article
Effect of Concentrations of Bacterial Consortia in Culture Medium from Wastewater in Microbial Fuel Cells
Department of Chemical Engineering, Faculty of Engineering, Universitas Indonesia, Kampus UI, 16424 Depok, Indonesia
Tania Surya Utami
Department of Chemical Engineering, Faculty of Engineering, Universitas Indonesia, Kampus UI, 16424 Depok, Indonesia
LiveDNA: 62.20122
Astry Eka Citrasari
Department of Chemical Engineering, Faculty of Engineering, Universitas Indonesia, Kampus UI, 16424 Depok, Indonesia
Heri Hermansyah
Department of Chemical Engineering, Faculty of Engineering, Universitas Indonesia, Kampus UI, 16424 Depok, Indonesia