ABSTRACT
The presence of textile dyes in wastewater of textile factories represent a major environment problem threatening the aquatic life. The decolorization of such harmful products is the major field of interest in research. In this study, Aspergillus niger, a brown rot fungi, isolated from the soil samples around the textile distillery industries of Al-Mahala Al-kobra, Egypt was evaluated for its efficiency in decolorization of different textile dyes; reactive red 198 (RR 198), reactive orange 122 (RO 122), reactive yellow 160 (RY 160), reactive blue 21 (RB 21) and reactive blue 19 (RB 19). Aspergillus niger showed a strong ability to decolorize various reactive dyes (azo, phthalocyanine and anthraquinone dyes) after 4 days. The decolorization was effective in an acidic environment (pH 4). Aspergillus niger grew well in a high concentration of dyes (500 mg L–1), resulting approximately in 100% decolorization extent in 5 days with RB 21, RB 19, RR 198 and RO 122 and could tolerate up to 1000 mg L–1 of dyes. The decolorization was more effective in the presence of sucrose. The Reactive Yellow 160, a diazo dye, was more resistant to decolorization by Aspergillus niger than the other dyes. High decolorization extent and facile conditions showed the potential for the Aspergillus niger to be used in biological treatment of textile dyes.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jest.2016.149.156
URL: https://scialert.net/abstract/?doi=jest.2016.149.156
INTRODUCTION
Synthetic dyes, which are extensively used in the textile industry, represent a major environmental problem. Due to inefficiency of the industrial dyeing process, 10-15% of the dyes are lost in the effluents of textile units, rendering them highly colored (Vaidya and Date, 1982; Boer et al., 2004). It is estimated that 280,000 t of textile dyes are discharged in such industrial effluents every year worldwide (Maas and Chaudhari, 2005).
Direct discharge of these effluents causes formation of toxic aromatic amines under anaerobic conditions in receiving media. In addition to their visual effect and their adverse impact in terms of Chemical Oxygen Demand (COD), many synthetic dyes are toxic, mutagenic and carcinogenic (Chung and Stevens, 1993). The efficient removal of dyes from textile industry effluents is still a major environmental challenge (Baldrian and Gabriel, 2003).
The commonly used dyes by the textile industry are azo dyes (reactive red 198, reactive yellow 160 and reactive orange 12), anthroquinone (reactive blue 19) and copper phthalocyanine (reactive blue 21). Degradation of these dyes, especially azo dyes, which comprise about 70% of all dyes used, is difficult due to their complex structure and synthetic nature (Swamy and Ramsay, 1999; Maas and Chaudhari, 2005). Also, the possible contamination of drinking water supplies is of concern because azo dyes are known to be enzymatically degraded in the human digestive system producing carcinogenic substances (Sheth and Dave, 2009).
Currently, various chemical, physical and biological treatment methods are used to remove color (Pala and Toket, 2002; Zhang et al., 2003). Because of the high cost and disposal problems, most of the chemical and physical methods for treating dye wastewater were not widely applied in the textile industries (Robinson et al., 2001; Mazmanci and Unyayar, 2005). Because synthetic dyestuffs are resistant to biological degradation, color removal by bioprocesses is also difficult (Shaul et al., 1991; Willmott et al., 1998). Decolorization generally occurs by the adsorption of dyestuffs on bacteria, rather than oxidation in aerobic systems. Some bacteria can biodegrade dyestuffs by azo reductase activity. However, the effluent at the end of biotransformation of dyestuffs could be toxic (Chung and Stevens, 1993). These problems limit large-scale application of bacterial decolorization.
Several fungi are capable of mineralizing pollutant compounds through their highly oxidative and non-specific ligninolytic enzymes, which are also responsible for the decolorization and degradation of many different dyes (Dos Santos et al., 2004). The white rod fungi, members of the Basidiomycetes, offer significant advantages over bacteria as Phanerochaete chrysosporium (Arora and Chander, 2004; Martins et al., 2002) and Trametes versicolor (Ramsay and Nguyen, 2002) are extremely efficient for textile dye degradation. Their extracellular enzymes which are non-specific can attack a wide variety of complex aromatic dyestuffs (Boer et al., 2004; Kamitsuji et al., 2005). Recently, there is a growing interest in studying the brown rot fungi; Aspergillus niger, for the decolorization and degradation of many different dyes because their biomass can be used as an adsorbent and serve as a part of a technical solution in water pollution control (Fu and Viraraghavan, 2000; Srividhya et al., 2012).
The present study aimed at using a newly screened, Aspergillus niger, a brown rot fungi, isolated from soil polluted with textile industry for decolorization of five reactive dyes. Various conditions required for decolorized have been optimized.
MATERIALS AND METHODS
Isolation and selection of decolorizing fungi: The soil samples were collected near the places where the effluents are discharged from textile factories in El-Mahalah El-kubra, Egypt.
Soil samples were subjected to serial dilutions and inoculaed onto the sterile dishes which consist of sterile Czapex-Dox agar medium and then we followed the spread plate method and kept the plates for incubation at 30°C for 4 days. Fungal colonies were identified by Department of Plant Diseases, Faculty of Agriculture Mansoura, Egypt.
Chemicals: The dyes used in the investigation were; three azo dyes (reactive red 198, reactive yellow 160 and reactive orange 122), one phthalocyanine dye (reactive blue 21) and one anthraouinone dye (reactive blue 19). The dyes were chosen according to their wide use in the textile industry in Al-Mahla Al-kubra, Egypt. Solutions of these dyes were prepared by dissolving the dyes in distilled water.
Culture conditions: Fifty milliliters of Czapex-Dox medium amended with 200 mg L–1) concentration of each of the tested textile dyes were transferred to 250 mL Erlenmeyer flasks. The flasks were inoculated with 2% (v/v) fungal spore suspension containing 3.2×106 CFU mL–1. The flasks were incubated at shaking conditions 150 rpm at 30°C for 5 days. Control experiments were performed under the same conditions described above but without fungi.
Effects of different parameters, including pH (3-8), dye concentrations (100, 200, 300, 400, 500 and 1000 mg L–1), sucrose concentrations (0, 5, 10, 15, 20, 25 and 30 g L–1), on dye decolorization were investigated. Each experiment was carried out in triplicate.
Decolorization: Samples were centrifuged at 10000x g for 10 min. The decolorization was determined by measuring the absorbance of the culture supernatant. For RB 21 the absorbance was observed at 664 nm, for RB 4 it was at 592 nm, for RR 198 it was at 550 nm, for RY 160 it was at 413 nm and for RO 122 it was at 493 nm. The color removal was calculated using the following equation:
where, Ab is the initial absorbance and Aa is the observed absorbance of cultivation.
RESULTS AND DISCUSSION
Isolation and selection of decolorizing fungus: The soil samples collected from several textile industries of Al-Mahla Al-kubra, were screened for fungi. About ten deferent morphologically distinct fungi were isolated from the soil. Among them two fungal isolates showed higher decolorization and they were identified as Aspergillus niger and Aspergillus oryzae. Aspergillus species are well adapted to textile wastewater and are frequently isolated from effluents and dye contaminated soils. Namdhari et al. (2012) and Gnanadoss and Jebapriya (2013), showed that fungal biomass (A. niger and A. nidulans) could effectively be used as an alternative to the conventional physic-chemical methods.
Effect of time course on decolorization: Time dependent decolorization for five dyes (200 g L–1) was studied (Fig. 1), the five dyes were decolorized after 4 days incubation. The dyes were absorbed by the biomass and the visual observation of decolorization of the dyes was from the 3rd-6th day of incubation Fig. 2. The color of the biomass of fungus changed to the color of the tested dye. The color on the biomass was reduced gradually from the 6th until 10th day of incubation. Bergsten-Torralba et al. (2009) and Yu and Wen (2005) reported that the decolorization of dyes by yeast and fungi can be due to adsorption of the dye to microbial cells or to biodegradation.
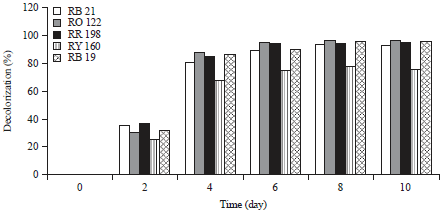 | |
| Fig. 1: | Decolorization of reactive dyes by Aspergillus niger after 0, 2, 4, 6, 8 and 10 days of incubation |
 | |
| Fig. 2(a-g): | Visual observation of decolorization of the dyes (a) RY 160, (b) RO 122, (c) RB 21 and (d) RR 198 by Aspergillus niger after 4 days (200 mg L–1) dye, (e and f) Decolorized culture supernatant after 4 days and (g) Biomass visually had adsorbed most of the dyes |
The present experiments demonstrated the efficiency of A. niger to decolorize different kinds of dyes, monoazo dyes (reactive red 198 and reactive orange 122), a diazo dye (reactive yellow 160), a phthalocyanine dye (reactive blue 21) and an anthroquinone dye (reactive blue 19) with differences in the decolorization ability. The mechanism of decolorization may be due to biosorbtion, which is dependent on functional groups in the dye molecule and in fungal biomass, which may also play role in the biosorption of dye (Raju et al., 2007; Fu and Viraraghavan, 2002). The fourth dye RY 160 (diazo dye) was more resistant to decolorization by A. niger than the other four tested dyes (Fig. 1), possibly due to its more complex molecular structure.
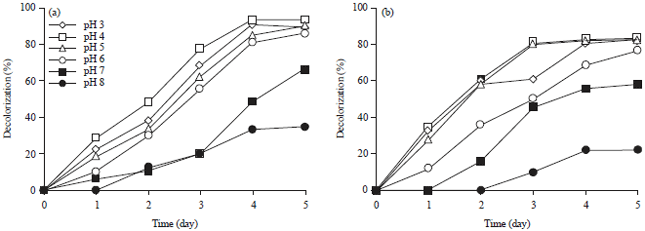 | |
| Fig. 3(a-b): | Effect of initial pH on decolorization of (a) Reactive blue 21 and (b) Reactive yellow 160 by A. niger |
Effect of initial pH on decolorization: The effect of initial pH on the decolorization of dyes by A. niger is shown in Fig. 3a and b. The results reveal that the organism was capable of decolorizing dyes over pH range of 3.0-8.0. The best decolorization was achieved at pH 3 and 4 in 4 days. This could be due to the fact that the optimum pH for the growth of A. niger was acidic. Similar decolorization extent was observed at pH 5 and 6 after 5 days. Whereas the rate of color removal was much lower at strongly alkaline condition (pH 8) and the decolorization process started after 2 days. The optimal decolorization of five dyes was achieved at pH 4 with (93.5, 97.0, 98.5, 83.0 and 96.7%) decolorization, for (RB 21, RO 122, RR 198, RY 160 and RB 19), respectively. The decolorization (%) of RY 160 was slightly lower than the other dyes (Fig. 3a). Most previous reports indicated pH optima in the range 3.0-5.0 in the case of T. vercicolor, Trichoderma sp., P. chrysosporium and Aspergillus fumigates (Mehna and Bajpai, 1995; Ramya et al., 2007; Akar et al., 2009).
These results showed that A. niger could decolorize reactive dyes in relatively wide range of pH, this makes the fungus suitable for practical treatment of dyes.
Effect of initial dye concentration on decolorization: With the increase of the initial dye concentration, the decolorization extent over the same time interval decreased. When the effect of different initial dye concentrations on decolorization was observed using 100, 200, 300, 400, 500 and 1000 mg L–1, the decolorization rate was increased by the increasing of dye concentration from 100-500 mg L–1 and the required time to reach a maximum decolorization extent was increased (Fig. 2a). The maximum decolorization was about 100% for (RB 21, RO 122, RR 198, RB 19) at 500 mg L–1 dye after 5 days.
However, at 1000 mg L–1, dye decolorization decreased sharply (47.5, 34.6, 46.3 and 45.8%) for RB 21, RO 122, RR 198, RB 19, respectively. These results were observed with all tested dyes except (RY 160), A. niger could decolorize RY 160 well at concentration 100 mg L–1, decolorization reaching 87%. However, above this concentration, dye decolorization decreased sharply, at 1000 mg L–1 only about 11.5% decolonization was achieved by A. niger (Fig. 2b). In addition the growth of fungi was inhibited at dye concentration 1000 mg L–1 (data not shown). Ramya et al. (2007) and Namdhari et al. (2012), showed that the higher concentration dye may be toxic to metabolite activity. This suggested that for A. niger the maximal dye easily treated concentration was 500 mg L–1 for four tested textile dyes and 100 mg L–1 for reactive yellow 160 (RY 160) (Fig. 4).
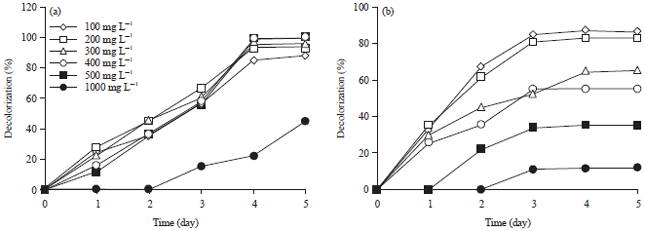 | |
| Fig. 4(a-b): | Effect of initial dye concentration on decolorization of (a) Reactive blue 21 and (b) Reactive Yellow 160 by Aspergillus niger |
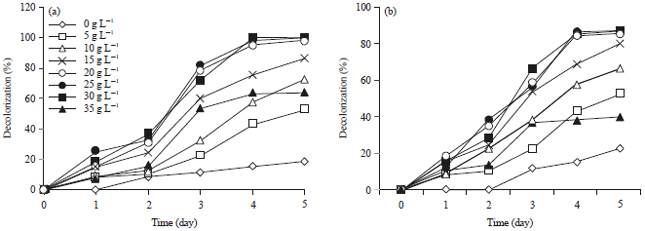 | |
| Fig. 5(a-b): | Effect of initial sucrose concentration on decolorization of (a) Reactive blue 21 and (b) Reactive yellow 160 by Aspergillus niger |
Effect of initial sucrose concentration on decolorization: The effect of initial sucrose concentration on decolorization by A. niger is shown in Fig. 3a and b. Addition of sucrose enhanced the decolorization of five dyes by A. niger. However lack of sucrose inhibited the decolorization activity of A. niger since only (35.7, 37.2, 42.4, 20.2 and 40.3%) decolorization was observed after 5 days for RB 21, RO 122, RR 198, RY 160, RB 19, respectively. In experiments with sucrose supplementation, A. niger exhibited strong decolorizing activity about 100% decolorization for four dyes and 88% for RY 160 extent in 4 days, except that when the sucrose concentration was 5 g L–1 the decolorization efficiency (18.5% in 5 days) was much lower (Fig. 5). The reason why low decolorization extent appeared when the sucrose concentration was 5 g L–1 may be that low sucrose concentration could not meet the growth requirements of the fungus. When the sucrose concentration was higher than 30 g L–1, the decolorization efficiency was much lower that may be due to that high sucrose concentration can inhibited the decolorizing activity (Chen et al., 2003; Ali et al., 2008). Present results showed that a certain concentration of carbon source (sucrose) was necessary for the A. niger decolorizing process.
CONCLUSION
In this study, Aspergillus niger was isolated from the soil samples around the textile distillery industries of Al-Mahala Al-kobra, Egypt showed high decolorizing activity against various reactive dyes (including azo, anthraquinone and phthalocyanine dyes) commonly used in the textile industries. Aspergillus niger could tolerate high concentrations of dyes (up to 1000 mg L–1), it proposed that A. niger has a practical application potential in the treatment of various dye effluents.
REFERENCES
- Akar, S.T., T. Akar and A. Cabuk, 2009. Decolorization of a textile dye, reactive red 198 (rr198), by Aspergillus parasiticus fungal biosorbent. Braz. J. Chem. Eng., 26: 399-405.
CrossRefDirect Link - Ali, N., Ikramullah, G. Lutfullah, A. Hameed and S. Ahmed, 2008. Decolorization of acid red 151 by Aspergillus niger SA1 under different physicochemical conditions. World J. Microbiol. Biotechnol., 24: 1099-1105.
CrossRefDirect Link - Arora, D.S. and M. Chander, 2004. Decolourisation of diverse industrial dyes by some Phlebia spp. and their comparison with Phanerochaete chrysosporium. J. Basic. Microbiol., 44: 331-338.
CrossRefDirect Link - Baldrian, P. and J. Gabriel, 2003. Lignocellulose degradation by Pleurotus ostreatus in the presence of cadmium. FEMS Microbiol. Lett., 220: 235-240.
CrossRefPubMedDirect Link - Bergsten-Torralba, L.R., M.M. Nishikawa, D.F. Baptista, D.P. Magalhaes and M. da Silva, 2009. Decolorization of different textile dyes by Penicillium simplicissimum and toxicity evaluation after fungal treatment. Braz. J. Microbiol., 40: 808-817.
PubMedDirect Link - Boer, C.G., L. Obici, C.G.M. de Souza and R.M. Peralta, 2004. Decolorization of synthetic dyes by solid state cultures of Lentinula (Lentinus) edodes producing manganese peroxidase as the main ligninolytic enzyme. Bioresour. Technology, 94: 107-112.
CrossRefDirect Link - Chen, K.C., J.Y. Wu, D.J. Liou and S.C.J. Hwang, 2003. Decolorization of the textile dyes by newly isolated bacterial strains. J. Biotechnol., 101: 57-68.
CrossRefDirect Link - Chung, K.T. and S.E. Stevens Jr., 1993. Degradation azo dyes by environmental microorganisms and helminths. Environ. Toxicol. Chem., 12: 2121-2132.
CrossRefDirect Link - Dos Santos, A.Z., J.M.C. Neto, C.R.G. Tavares and S.M.G. da Costa, 2004. Screening of filamentous fungi for the decolorization of a commercial reactive dye. J. Basic Microbiol., 44: 288-295.
CrossRefDirect Link - Fu, Y. and T. Viraraghavan, 2000. Removal of a dye from an aqueous solution by the fungus Aspergillus niger. Water Qual. Res. J. Can., 35: 95-111.
Direct Link - Fu, Y. and T. Viraraghavan, 2002. Dye biosorption sites in Aspergillus niger. Biores. Technol., 82: 139-145.
CrossRefDirect Link - Gnanadoss, J.J. and G.R. Jebapriya, 2013. Decolourization of synthetic dyes using free and immobilized Aspergillus species. Res. Biotechnol., 5: 20-23.
Direct Link - Kamitsuji, H., Y. Honda, T. Watanabe and M. Kuwahara, 2005. Mn2+ is dispensable for the production of active MnP2 by Pleurotus ostreatus. Biochem. Biophys. Res. Commun., 327: 871-876.
CrossRefPubMedDirect Link - Martins, M.A.M., M.J. Queiroz, A.J.D. Silvestre and N. Lima, 2002. Relationship of chemical structures of textile dyes on the pre-adaptation medium and the potentialities of their biodegradation by Phanerochaete chrysosporium. Res. Microbiol., 153: 361-368.
CrossRefPubMedDirect Link - Maas, R. and S. Chaudhari, 2005. Adsorption and biological decolourization of azo dye Reactive Red 2 in semicontinuous anaerobic reactors. Process Biochem., 40: 699-705.
CrossRefDirect Link - Mazmanci, M.A. and A. Unyayar, 2005. Decolourisation of Reactive Black 5 by Funalia trogii immobilised on Luffa cylindrica sponge. Process Biochem., 40: 337-342.
CrossRefDirect Link - Mehna, A., P. Bajpai and P.K. Bajpai, 1995. Studies on decolorization of effluent from a small pulp mill utilizing agriresidues with Trametes versicolor. Enzyme Microb. Technol., 17: 18-22.
CrossRefDirect Link - Namdhari, B.S., S.K. Rohilla, R.K. Salar, S.K. Gahlawat, P. Bansal and A.K. Saran, 2012. Decolorization of Reactive Blue MR, using Aspergillus species isolated from textile waste water. ISCA J. Biol. Sci., 1: 24-29.
Direct Link - Pala, A. and E. Tokat, 2002. Color removal from cotton textile industry wastewater in an activated sludge system with various additives. Water Res., 36: 2920-2925.
CrossRefDirect Link - Ramya, M., B. Anusha, S. Kalavathy and S. Devilaksmi, 2007. Biodecolorization and biodegradation of Reactive Blue by Aspergillus sp. Afr. J. Biotechnol., 6: 1441-1445.
Direct Link - Raju, N.S., G.V. Venkataramana, S.T. Girish, V.B. Raghavendra and P. Shivashankar, 2007. Isolation and evaluation of indigenous soil fungi for decolourization of textile dyes. J. Applied Sci., 7: 298-301.
CrossRefDirect Link - Ramsay, J.A. and T. Nguyen, 2002. Decoloration of textile dyes by Trametes versicolor and its effect on dye toxicity. Biotech. Lett., 24: 1757-1761.
CrossRefDirect Link - Robinson, T., G. McMullan, R. Marchant and P. Nigam, 2001. Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Bioresour. Technol., 77: 247-255.
CrossRefDirect Link - Shaul, G.M., T.J. Holdsworth, C.R. Dempsey and K.A. Dostal, 1991. Fate of water soluble azo dyes in the activated sludge process. Chemosphere, 22: 107-119.
CrossRefDirect Link - Sheth, N.T. and S.R. Dave, 2009. Optimisation for enhanced decolourization and degradation of Reactive Red BS C.I. 111 by Pseudomonas aeruginosa NGKCTS. Biodegradation, 20: 827-836.
CrossRefDirect Link - Srividhya, R., A.L.H. Mary, Mukeshgoel and S. Rangabhashiyam, 2012. Decolorization study of synthetic optilan red dye by Aspergillus niger. Int. J. Recent Scient. Res., 3: 301-303.
Direct Link - Swamy, J. and J.A. Ramsay, 1999. The evaluation of white rot fungi in the decoloration of textile dyes. Enzyme Microb. Technol., 24: 130-137.
CrossRefDirect Link - Willmott, N., J. Guthrie and G. Nelson, 1998. The biotechnology approach to colour removal from textile effluent. J. Soc. Dyers Colourists, 114: 38-41.
CrossRefDirect Link - Yu, Z. and X. Wen, 2005. Screening and identification of yeasts for decolorizing synthetic dyes in industrial wastewater. Int. Biodeterior. Biodegrad., 56: 109-114.
CrossRefDirect Link - Zhang, S.J., M. Yang, Q.X. Yang, Y. Zhang, B.P. Xin and F. Pan, 2003. Biosorption of reactive dyes by the mycelium pellets of a new isolate of Penicillium oxalicum. Biotechnol. Lett., 25: 1479-1482.
CrossRefDirect Link








