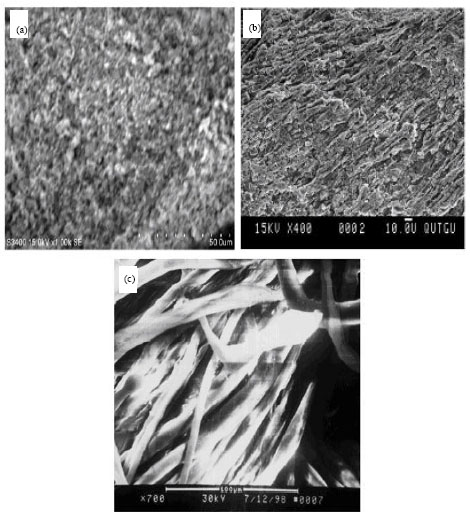
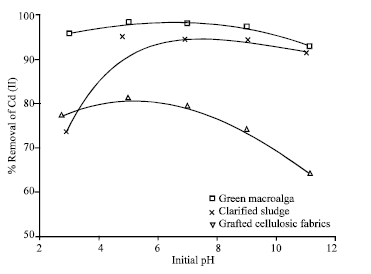
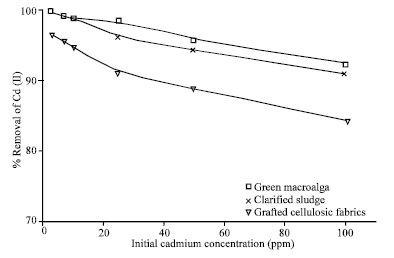
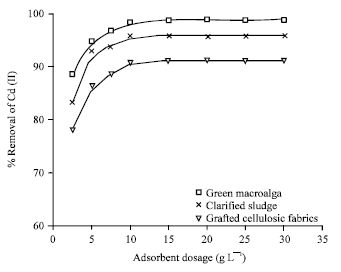
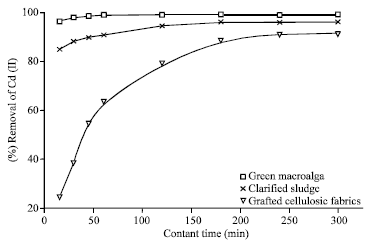
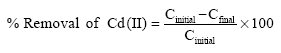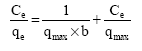Research Article
Comparative Study of the Removal of Cadmium from Aqueous Solution by using Low-cost Adsorbents
Unite de Recherche: Chimie Theorique et Reactivite, Faculte des Sciences de Monastir, 5019 Monastir, Tunisie
Y. Hannachi
Unite de Recherche: Chimie Theorique et Reactivite, Faculte des Sciences de Monastir, 5019 Monastir, Tunisie
A. Ghorbel
Unite de Recherche: Chimie Theorique et Reactivite, Faculte des Sciences de Monastir, 5019 Monastir, Tunisie
T. Boubaker
Unite de Recherche: Chimie Theorique et Reactivite, Faculte des Sciences de Monastir, 5019 Monastir, Tunisie