ABSTRACT
In the recent years, the complexation-ultrafiltration technique has been shown to be a promising technique for removal of heavy metals in solution. In this study, a polymer-enhanced ultrafiltration process has been investigated for removal of toxic heavy metals such as Cu (II), Ni (II) and Cr (III) from synthetic wastewater solutions. Carboxy methyl cellulose as a water-soluble polymer was used for complexing the cationic forms of the heavy metals before filtration. The functions of the metal-CMC complexation are to increase their molecular weight and their size. The size of the complex has to be larger than the pores of the selected membrane so the complex can be retained. Permeate water is then purified from the heavy metals. Filtration experiments were performed with ultrafiltration membrane system, equipped with a polyethersulfon membrane with a 10000 Daltons cut-off. The pressure was fixed at 1 bar with a permeate flow rate of 7.5 L h -1 . Different parameters, affecting the percentage rejection of the metals, such as pH and metal/CMC ratio have been investigated. Results obtained revealed that the maximum percentage of the metals rejection was achieved at pH ≥ 7 with increasing of the CMC concentration. Advantages of that technology over the other conventional technologies are the low energy requirements involved in ultrafiltration, the very fast reaction kinetics and the high selectivity of separation.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jest.2008.151.156
URL: https://scialert.net/abstract/?doi=jest.2008.151.156
INTRODUCTION
Heavy metal removal from inorganic effluent can be achieved by conventional treatment processes such as chemical precipitation, ion exchange and electrochemical removal. These processes have significant disadvantages, which are for instance incomplete removal, high-energy requirements and production of toxic sludge (Eccles, 1999). Recently, membrane separation processes such as; Ultrafiltration (UF), Nanofiltration (NF) and Reverse Osmosis (RO), have been increasingly used for the treatment of inorganic effluent to decrease the amount of wastewater produced and to improve the quality of the treated effluent. Complexation-ultrafiltration, proves to be a new promising technology for separation of heavy metals from effluents. The use of water soluble metal-binding polymers in combination with UF is a hybrid approach to concentrate selectively and to recover valuable elements as heavy metals. In the complexation-UF process cationic forms of heavy metals are first complexed by a macroligand in order to increase their molecular weight with a size larger than the pores of the selected membrane that can be retained whereas permeate water is then purified from the heavy metals (Rether and Schuster, 2003). The advantages of complexation-filtration process are the high separation selectivity due to the use of a selective binding and low-energy requirements involved in these processes. Water-soluble polymeric ligands have shown to be powerful substances to remove trace metals from aqueous solutions and industrial wastewater through membrane processes. However, the choice of the proper water-soluble macroligands is important for developing this technology. Several experiments have been done using biopolymer macroligands able to complex heavy metal ions such as chitosan (Juang and Shiau, 2000; Cervera et al., 2003; Llorens et al., 2004). The other group of macroligands is the synthetic macroligands such as carboxyl methyl cellulose (Petrov and Nenov, 2004), diethylaminoethyl cellulose (Trivunac and Stevanovic, 2006), polyvinylethylenimin (Muslehiddinoglu et al., 1998; Canizares et al., 2002), polyvinyl alcohol (Vieira et al., 2001), poly (acrilic acid) (Bodzek et al., 1999; Zhang and Xu, 2003). Removal of pollutants by micellarenhanced ultrafiltration was investigated by several authors (Baek et al., 2003; Juang et al., 2003; Aroua et al., 2007). This study will be focused on investigation the applicability of the complexation-ultrafiltration process for removal of toxic heavy metals, in particular; Cu (II), Ni (II) and Cr (III), from synthetic wastewater solutions. To highlight the metals removal performance, the main operating conditions such as pH, pollutants and ligand concentrations have been investigated.
MATERIALS AND METHODS
Materials
Standard metal solutions of Cu (II), Ni (II) and Cr (III) with metal concentration of 1000 mg L-1 were used as metal precursors. The complexation experiments were carried out with carboxymethyl cellulose (CMC, viscosity 25-75 mPa.s) cellulose as a water-soluble polymer. The pH of the aqueous solutions was adjusted using 1 M HCl and 1 M NaOH. All reagents were of analytical grade and supplied by Merck (Wiesbaden, Germany). The filtration experiments were performed with ultrafiltration membrane system, equipped with a polyethersulfon membrane (MOLSEP® Hohlfasermodule FUS 0181, NADIR Filtration GmbH, Germany) with a 10000 Daltons cut-off.
Complexation-Ultrafiltration Procedure
Solution of 5l containing different concentrations of the metal and CMC was stirred for metal-CMC complexation. The resulting solution was placed into the reactor system and the ultrafiltration was started. The pressure was fixed at 1 bar with a permeate flow rate of 7.5 L h-1. At the beginning of the filtration, both permeate and retenate were returned to the reactor tank (run in a cycling mode) in order to achieve steady-state operation. The solution pH was controlled and corrected using pH meter (WTW, ISA RWTH-AACHEN, Germany). After circulating the solution (cycle mode) for 10 min at constant pH, a sample was taken for metal analysis. Sequence runs of the permeate water (each of 1l every 8 min) were then collected in separate containers. Samples from the permeate solutions and the retenate were taken for metal analysis. At the end of the experiment, the membrane was immediately cleaned with warm water and regenerated successively by circulation for two runs with water solution (as prescribed by the membrane Company). In the first run, 1 g of sodium dithionite was added and the solution pH was adjusted to about 9 with 5% NaOH. In the second run, the solution was then neutralized with 5% citric acid.
Chemical Analysis
Metal concentrations, in the feed, permeate and retenate solutions, were analyzed by a photometer (HACH LANGE, LASA 100) with cuvette tests of LCK 329 for Cu (II), LCK 337 for Ni (II) and LCK 313 for Cr (III).
RESULTS AND DISCUSSION
Membrane Specifications
The specifications of the used ultrafiltration membrane are shown in Table 1. The Polyethersulfone membrane is hydrophilic and constructed from pure polyethersulfone polymer membrane. It is designed to remove particulates from solutions during general filtration
| Table 1: | Specifications of MOLSEP® Hohlfasermodule (FUS 0181) ultrafiltration membrane |
 | |
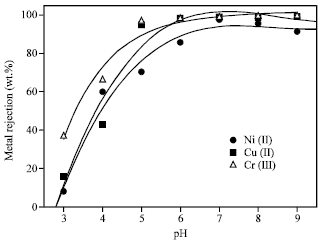 | |
| Fig. 1: | Effect of pH on the rejection of Ni (II), Cu (II) and Cr (III) from aqueous solutions using polyethersulfon ultrafiltration membrane (CMC = 1 g L-1, Metal ion conc. = 10 mg L-1, p = 1 bar) |
CMC-Metal Ions Complexes
CMC form polymer complexes with transition metal ions. The binding sites in Cr (III) and Ni (II) complexes were the etheroxygen of the hydroxyl group, while in Cu (II) complexes, the binding sites were the oxygen of ethoxyl groups and the primary alcoholic O atom of glucopyranose rings. These complexes would most likely exhibit an octahedral geometry with Cr (III) and Ni (II), but a square planar configuration in the Cu (II) complex (Norkus et al., 2002).
Effect of Solution pH
The effect of pH on the rejection of the metal ions from aqueous solutions using polyethersulfon ultrafiltration membrane is shown in Fig. 1. The CMC/Metal ratio was 100 and the applied pressure was 1 bar. It can be seen that the metal ions rejection efficiency was increased with pH increase, reaching maxima values at pH ≥7. This can be attributed to that, the stability of the formed M-CMC complexes is pH dependent. At low pH, the affinity of CMC towards the metal ions is weak due to the presence of positive charges and so the stability of the complex is low (Fig. 1). As pH increases, the affinity and stability of CMC-metal complexes increases. The metal rejection efficiency values were 97.6, 99.1 and 99.5% for Cu (II), Ni (II) and Cr (III) ions, respectively at pH 7.
Effect of CMC/Metal Ratio
Figure 2 shows the effect of CMC/M ratio on the complexation and filtration processes. It is clear that increasing the CMC ratio enhanced the metal bonding process. This is due to that, the molecules of the metal hydroxyl complexes with CMC particles start to agglomerate forming bigger particles that are rejected by the membrane. The maximum metal rejection was achieved at CMC/M ratio of 50.
Effect of Metal Ion Concentration
Almost all the metal ions were rejected at low metal ions concentration (10 mg L-1). On increasing the metal ions concentration, the membrane had been worked efficiently on a wide range of
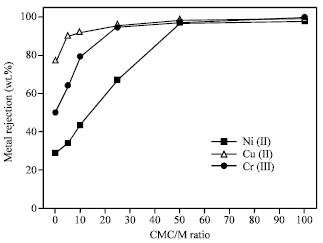 | |
| Fig. 2: | Effect of CMC concentration on the rejection of Ni (II), Cu (II) and Cr (III) from aqueous solutions using polyethersulfon ultrafiltration membrane (pH 7, Metal ion conc. = 10 mg L-1, p = 1 bar) |
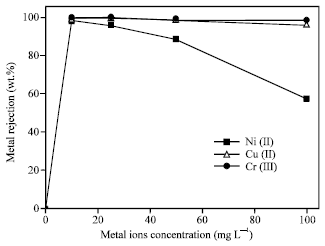 | |
| Fig. 3: | Effect of metal ions concentration on the rejection of Ni (II), Cu (II) and Cr (III) from aqueous solutions using polyethersulfon ultrafiltration membrane (pH 7, CMC = 1 g L-1, p = 1 bar) |
concentration up to 100 mg L-1 for both of Cu (II) and Cr (III) ions (Fig. 3). However, with Ni (II) ions, the metal rejection decreased gradually with increase of Ni (II) ion concentration, the metal rejection efficiency was 57% at Ni (II) ion concentration of 100 mg L-1. This is due to that CMC shows a high specific affinity towards certain cations. According to Irving-Williams series, which ranks the divalent metals ions in their tendency to complexity, regardless of the nature of the complexing agent, the cations were ranked as follows: Cu (II) > Zn (II) > Pb (II) > Ni (II) (Petro and Nenov, 2004). The current results agreed and confirmed that behavior for the divalent cations. The order of metal rejection was that Cu (II) > Cr (III) >> Ni (II). At high metal ions concentration, Ni (II) ions are partially bound to the CMC surface and so the rejection efficiency decreases.
Metals Rejection in Aqueous Solution Containing the Metal Ions Simultaneously
Metals rejection in aqueous solution containing Ni (II), Cu (II) and Cr (III) ions simultaneously was investigated. The results show the similarity in the efficiency of metals rejection in both of the individuals and the simultaneous solutions (Table 2).
| Table 2: | Metals rejection in both of individuals and simultaneous solutions |
| | |
| (pH 7, CMC = 1 g L?1, Metal ion conc. = 25 mg L?1, p = 1 bar) | |
CONCLUSIONS
Complexation-ultrafiltration process has been successfully applied for removal of Cu (II), Ni (II) and Cr (III) ions from synthetic wastewater solutions. Polyethersulfon (FUS 0181) was used as ultrafiltration membrane, while carboxy methyl cellulose was used as a metal complexing agent. It was shown that the compleaxtaion and filtration processes are pH dependent, the metal rejection was more efficient at neutral and alkaline conditions than at acidic one. The metal rejection efficiency values were 97.6, 99.1 and 99.5% for Cu (II), Ni (II) and Cr (III) ions, respectively at pH 7. At high metal ions concentration, the sequence with regards to complexing abilities of CMC was Cu (II) > Cr (III) >> Ni (II). The process is characterized by low energy requirements involved in the ultrafiltration, the very fast reaction kinetics and the high selectivity of separation.
REFERENCES
- Aroua, M.K., F.M. Zuki and N.M. Sulaiman, 2007. Removal of chromium ions from aqueous solutions by polymer-enhanced ultrafiltration. J. Hazard. Mater., 147: 752-758.
Direct Link - Baek, K., B.K. Kim, H.J. Cho and J.W. Yang, 2003. Removal characteristics of anionic metals by micellar-enhanced ultrafiltration. J. Hazard. Mater., 99: 303-311.
CrossRef - Bodzek, M., I. Korus and K. Loska, 1999. Application of the hybrid complexation-ltrafiltration process for removal of metal ions from galvanic wastewater. Desalination, 121: 117-121.
CrossRef - Canizares, P., A. Perez and R. Camarillo, 2002. Recovery of heavy metals by means of ultrafiltration with water-soluble polymers: Calculation of design parameters. Desalination, 144: 279-285.
CrossRef - Cervera, M.L., M.C. Arnal and M. de la Guardia, 2003. Removal of heavy metals by using adsorption on alumina or chitosan. Anal. Bioanal. Chem., 375: 820-825.
CrossRefDirect Link - Eccles, H., 1999. Treatment of metal-contaminated wastes: Why select a biological process? Trends Biotechnol., 17: 462-465.
CrossRef - Juang, R.S. and R.C. Shiau, 2000. Metal removal from aqueous solutions using chitosan-enhanced membrane filtration. J. Membr. Sci., 165: 159-167.
CrossRef - Juang, R.S., Y.Y. Xu and C.L. Chen, 2003. Separation and removal of metal ions from dilute solutions using micellar-enhanced ultrafiltration. J. Membr. Sci., 218: 257-267.
CrossRef - Llorens, J., M. Pujola and J. Sabate, 2004. Separation of cadmium from aqueous streams by polymer enhanced ultrafiltration: A two-phase model for complexation binding. J. Membr. Sci., 239: 173-181.
CrossRef - Muslehiddinoglu, J., Y. Uludag, H.O. Ozbelge and L. Yilmaz, 1998. Effect of operating parameters on selective separation of heavy metals from binary mixtures via polymer enhanced ultrafiltration. J. Membr. Sci., 140: 251-266.
CrossRef - Norkus, E., J. Vaiciuniene and D. Virbalyte, 2002. Interaction of copper (II) with cellulose pulp. Chemija (Vilnius), T13: 75-84.
Direct Link - Petrov, S. and V. Nenov, 2004. Removal and recovery of copper from wastewater by a complexation-ultrafiltration process. Desalination, 162: 201-209.
CrossRef - Rether, A. and M. Schuster, 2003. Selective separation and recovery of heavy metal ions using water-soluble N-benzoylthiourea modified PAMAM polymers. Reactive Functional Polymers, 57: 13-21.
CrossRef - Trivunac, K. and S. Stevanovic, 2006. Removal of heavy metal ions from water by complexation-assisted ultrafiltration. Chemosphere, 64: 486-491.
CrossRef - Vieira, M., C.R. Tavares, R. Bergamasco and J.C.C. Petrus, 2001. Application of ultrafiltration-complexation process for metal removal from pulp and paper industry wastewater. J. Membr. Sci., 194: 273-276.
CrossRef - Zhang, Y.F. and Zh.L. Xu, 2003. Study on the treatment of industrial wastewater containing Pb2+ ion using a coupling process of polymer complexation-ultrafiltration. Separ. Sci. Technol., 38: 1585-1596.
CrossRefDirect Link









Prof.Dr.Ahmed Moosa Reply
Very well Done and very informative