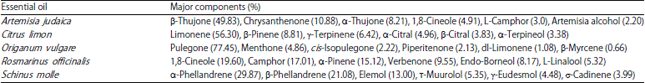
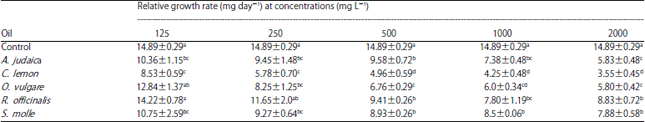
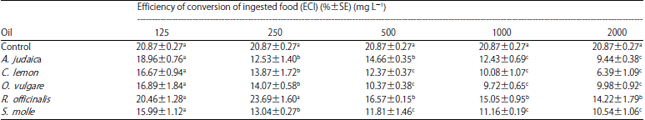
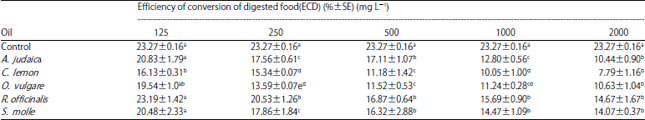
Research Article
Effects of Essential Oils on Growth, Feeding and Food Utilization of Spodoptera littoralis Larvae
Department of Applied Entomology and Zoology, Faculty of Agriculture, 21545 El-Shatby, Alexandria University, Alexandria, Egypt
Hossam El-Din Zahran
Department of Applied Entomology and Zoology, Faculty of Agriculture, 21545 El-Shatby, Alexandria University, Alexandria, Egypt
Samir Abdelgaleil
Department of Pesticide Chemistry and Technology, Faculty of Agriculture, 21545 El-Shatby, Alexandria University, Alexandria, Egypt
LiveDNA: 20.23527