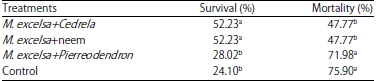Research Article
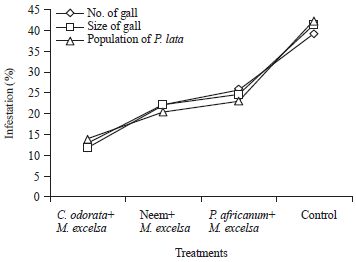
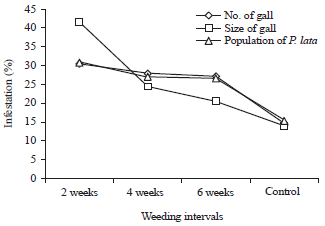
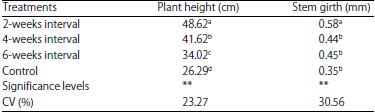
Evaluation of Mixed Planting and Weeding Regime for the Control of Iroko Gall Bug on Milicia excelsa
Federal College of Forestry, Forestry Research Institute of Nigeria, PMB 5087, Jericho, Ibadan, Oyo State, Nigeria
LiveDNA: 234.5685
Adebayo Amos Omoloye
Department of Crop Protection and Environmental Biology, University of Ibadan, Ibadan, Nigeria