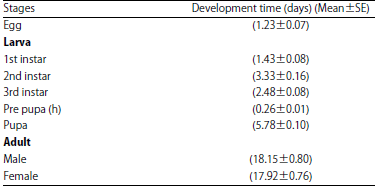
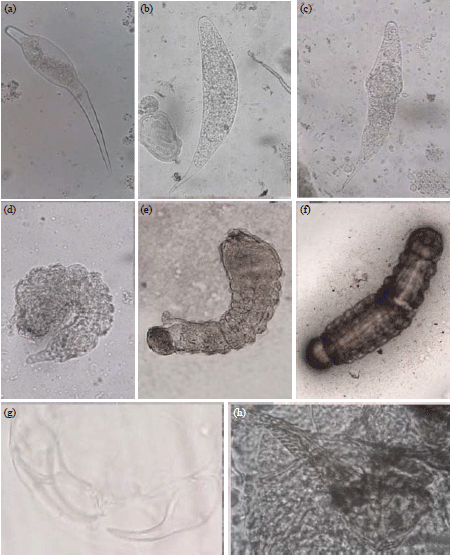
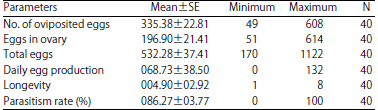
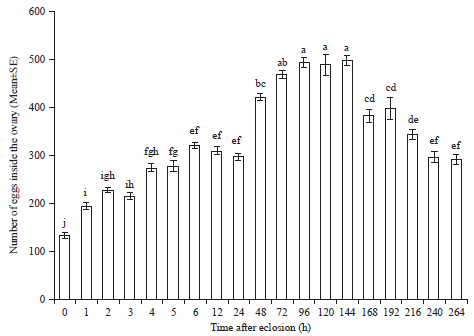
Research Article
Biology and Fitness Characteristics of Apanteles taragamae Viereck (Hymenoptera: Braconidae)
Laboratory of Biological Control, Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Jalan Kamper Kampus, IPB Dramaga, 16680 Bogor, Indonesia
Nurul Novianti
Laboratory of Biological Control, Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Jalan Kamper Kampus, IPB Dramaga, 16680 Bogor, Indonesia
Pudjianto
Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Jalan Kamper Kampus, IPB Dramaga, 16680 Bogor, Indonesia
Syafrida Manuwoto
Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Jalan Kamper Kampus, IPB Dramaga, 16680 Bogor, Indonesia
Damayanti Buchori
Department of Plant Protection, Faculty of Agriculture, Bogor Agricultural University, Jalan Kamper Kampus, IPB Dramaga, 16680 Bogor, Indonesia
LiveDNA: 62.16628