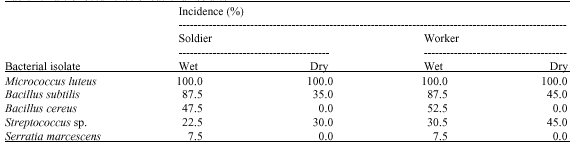
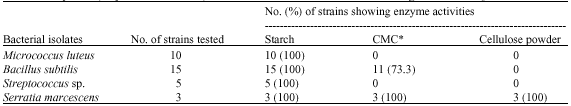
Research Article
Effects of Seasonal Changes on the Microflora In the Hindgut of Wood-Eating Termites
Department of Microbiology, University of Ado-Ekiti, P.M.B. 5363, Ado-Ekiti, Nigeria
E.Y. Aderibigbe
Department of Microbiology, University of Ado-Ekiti, P.M.B. 5363, Ado-Ekiti, Nigeria