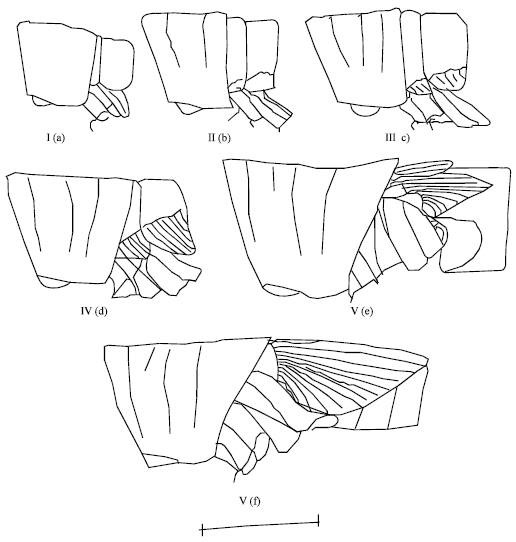
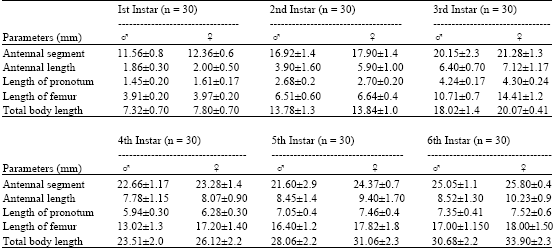
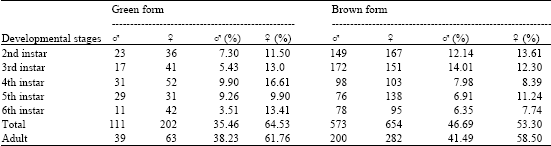
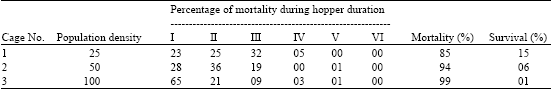
Research Article
Life History and Economic Importance of Hieroglyphus nigrorepletusBolivar (Hemiacridinae: Acrididae: Orthoptera) from Pakistan
Department of Zoology, University of Sindh, Jamshoro, Pakistan
M. Saeed Wagan
Department of Zoology, University of Sindh, Jamshoro, Pakistan