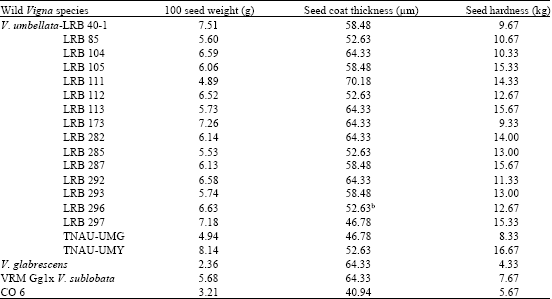
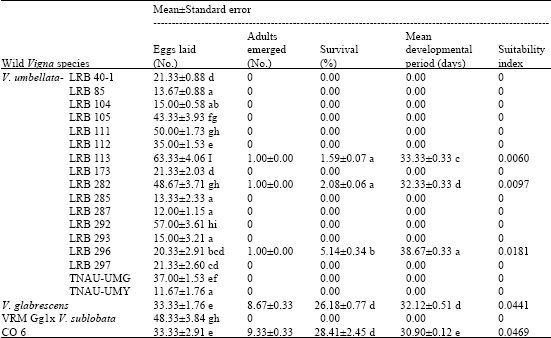
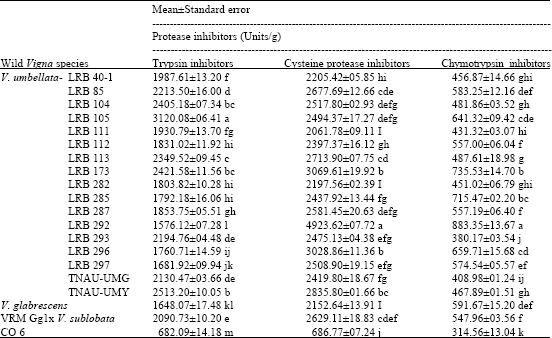
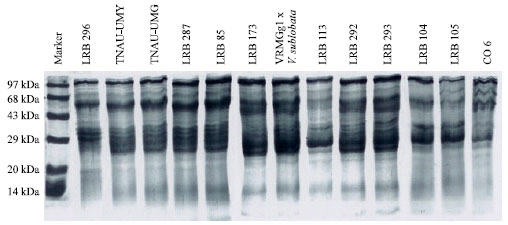
Research Article
Biochemical Basis of Resistance in Rice Bean, Vigna umbellata Thunb. (Ohwi and Ohashi) Against Callosobruchus maculatus F.
Department of Agricultural Entomology, Tamil Nadu Agricultural University, Coimbatore, 641 003, Tamil Nadu, India
C. Durairaj
Department of Pulses, Tamil Nadu Agricultural University, Coimbatore, 641 003, Tamil Nadu, India