Research Article
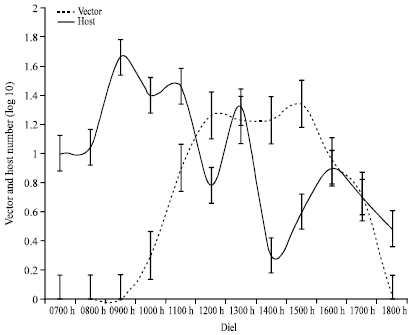
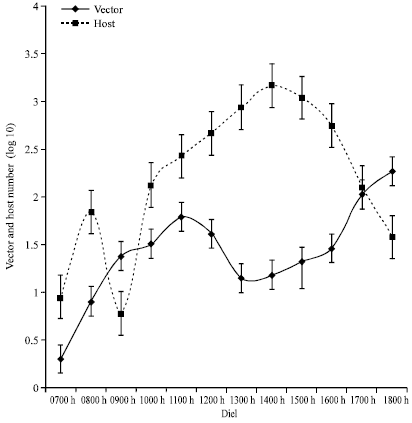
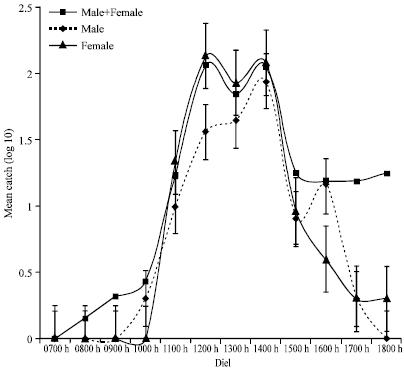
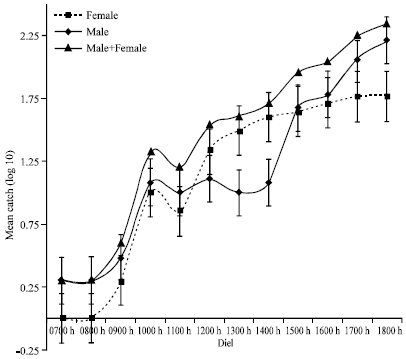
Diurnal Activity of Allopatric Populations of Glossina pallidipes and Hosts and their Implications on Risk of Transmission of Sleeping Sickness in Kenya
Trypanosomiasis Research Centre-Kenya Agricultural Research Institute, Box 362, Kikuyu Kenya
E.D. Kokwaro
School of Pure and Applied Sciences, Kenyatta University, Box 43844 Nairobi
J.M. Kiragu
Trypanosomiasis Research Centre-Kenya Agricultural Research Institute, Box 362, Kikuyu Kenya
G.A. Murilla
School of Pure and Applied Sciences, Kenyatta University, Box 43844 Nairobi