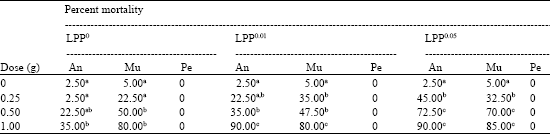
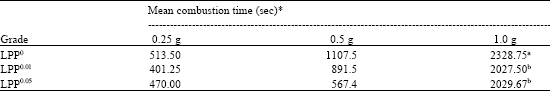Research Article
Insecticidal and Repellency Effects of Smoke from Plant Pellets with or Without D-allethrin 90 EC Against Three Medical Insects
Department of Zoology, Faculty of Science, Lagos State University, Lagos-Badagry Expressway, Ojo, P.M.B 1087 Apapa, Lagos, Nigeria
K. O. Teslim
Department of Zoology, Faculty of Science, Lagos State University, Lagos-Badagry Expressway, Ojo, P.M.B 1087 Apapa, Lagos, Nigeria
O. A. Fasasi
Department of Zoology, Faculty of Science, Lagos State University, Lagos-Badagry Expressway, Ojo, P.M.B 1087 Apapa, Lagos, Nigeria