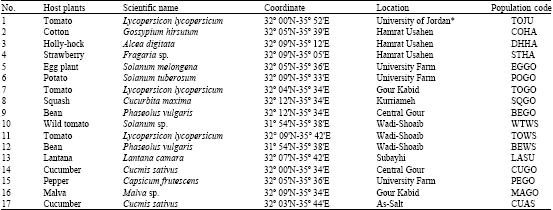
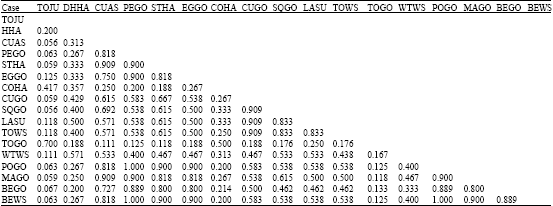
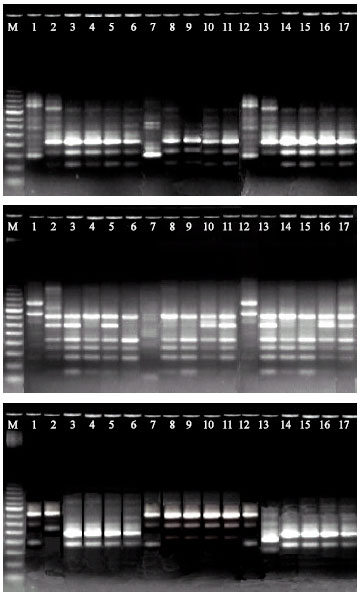
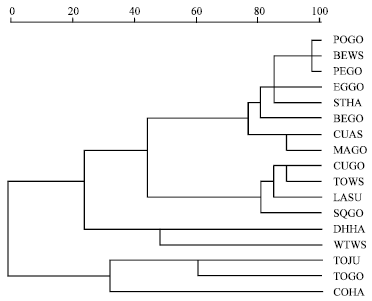
Research Article
Survey of Bemisia tabaci (Gennadius) (Homoptera: Aleyrodidae) Biotypes in Jordan using RAPD marker
Instructor in Plant Protection, Department of Agricultural Sciences, Al-Shobak University Collage, Al-Balqa Applied University, I 9I I 7-Salt-Jordan