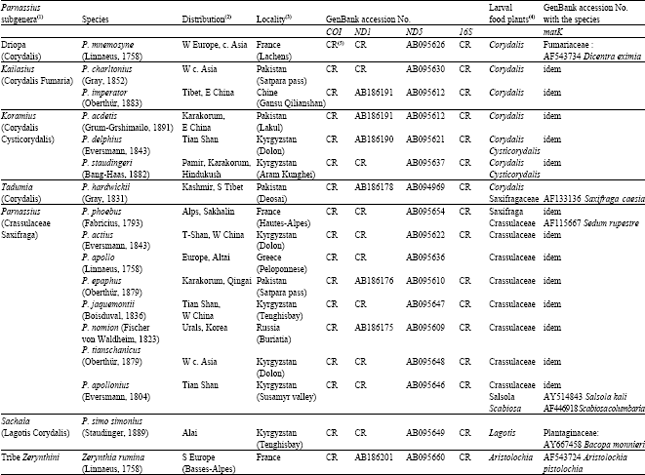
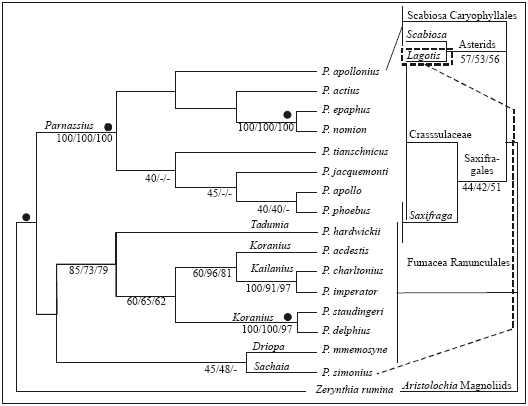
Research Article
Patterns of Speciation and Adaptive Radiation in Parnassius Butterflies
E.R. Biodiversity and Environment, Case 5, Pl. V. Hugo, Universite de Provence, 13331, Marseilles Cedex 3, France
F. Petenian
E.R. Biodiversity and Environment, Case 5, Pl. V. Hugo, Universite de Provence, 13331, Marseilles Cedex 3, France
E. Cosson
E.R. Biodiversity and Environment, Case 5, Pl. V. Hugo, Universite de Provence, 13331, Marseilles Cedex 3, France
E. Faure
E.R. Biodiversity and Environment, Case 5, Pl. V. Hugo, Universite de Provence, 13331, Marseilles Cedex 3, France