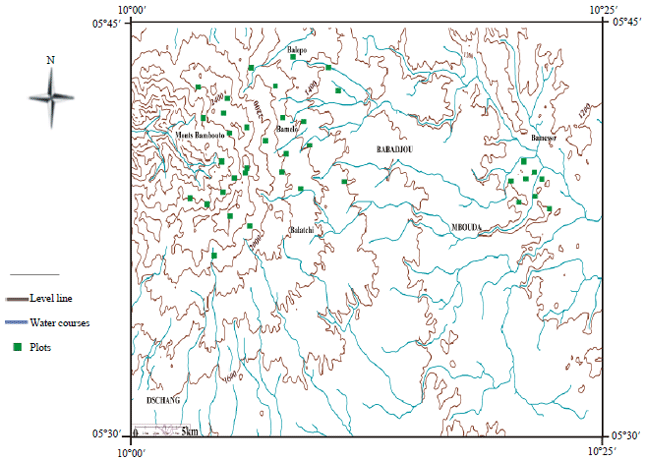
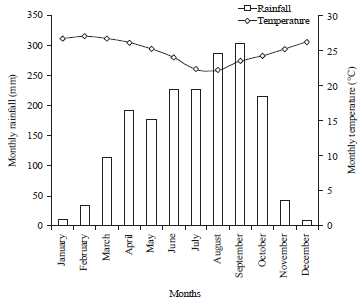
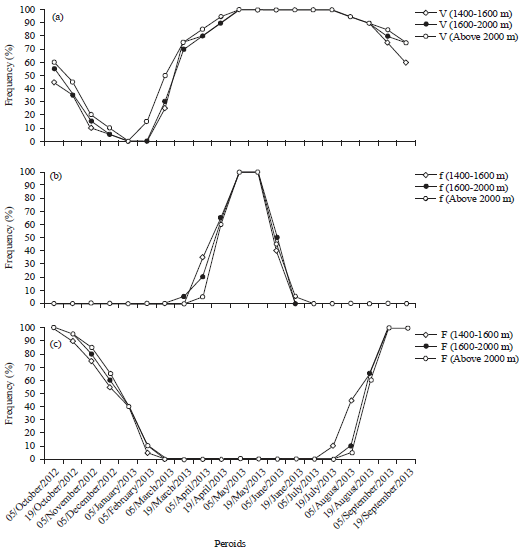
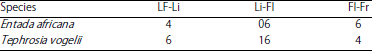Research Article
Phenological Behaviour of Tropical Tree Species in Three Altitudinal Zones of Bambouto Mountains, West Cameroon
Department of Plant Biology, Faculty of Sciences, University of Dschang, P.O. Box 67, Dschang, Cameroon
LiveDNA: 237.26373
Victor Francois Nguetsop
Department of Plant Biology, Faculty of Sciences, University of Dschang, P.O. Box 67, Dschang, Cameroon
Grace Mendi Anjah
Department of Plant Biology, Faculty of Sciences, University of Dschang, P.O. Box 67, Dschang, Cameroon
Marie Caroline Momo Solefack
Department of Plant Biology, Faculty of Sciences, University of Dschang, P.O. Box 67, Dschang, Cameroon
Walter Ndam Tacham
Department of Plant Biology, Faculty of Sciences, University of Dschang, P.O. Box 67, Dschang, Cameroon
Samuel Severin Kenfack Feukeng
Department of Plant Biology, Faculty of Sciences, University of Dschang, P.O. Box 67, Dschang, Cameroon