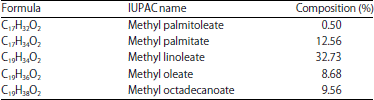
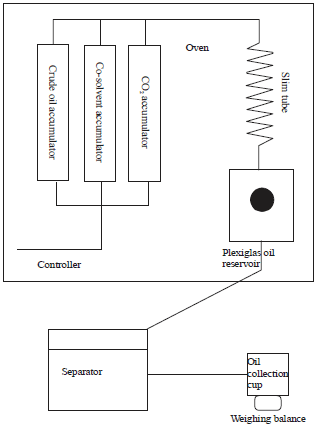
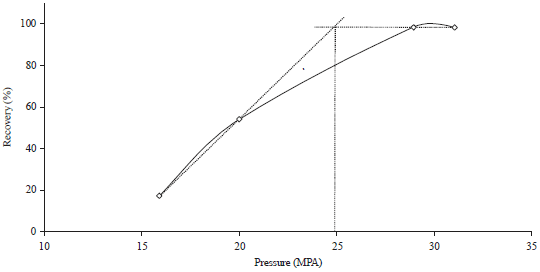
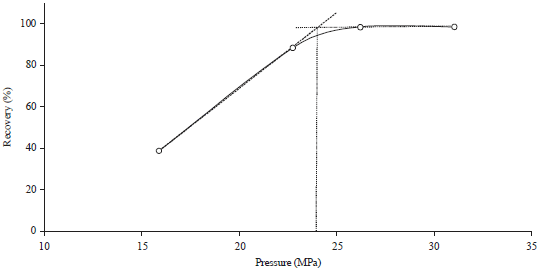
Research Article
Fatty Acid Methyl Ester from Rubber Seed Oil as Additives in Reducing the Minimum Miscibility Pressure of CO2 and Crude Oil
Centre of Enhanced Oil Recovery, Universiti Teknologi Petronas, 32610 Seri Iskandar, Perak, Malaysia
Mariyamni Awang
Centre of Enhanced Oil Recovery, Universiti Teknologi Petronas, 32610 Seri Iskandar, Perak, Malaysia
Suzana Yusup
Laboratory of Biomass Processing, Department of Chemical Engineering, Center of Biofuel Biochemical, Green Technology, Universiti Teknologi Petronas, 32610 Seri Iskandar, Perak, Malaysia