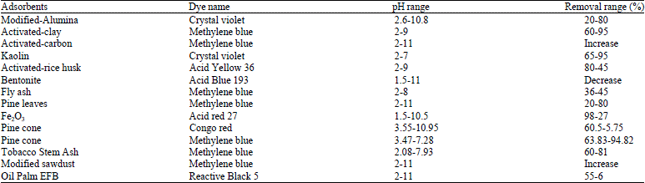
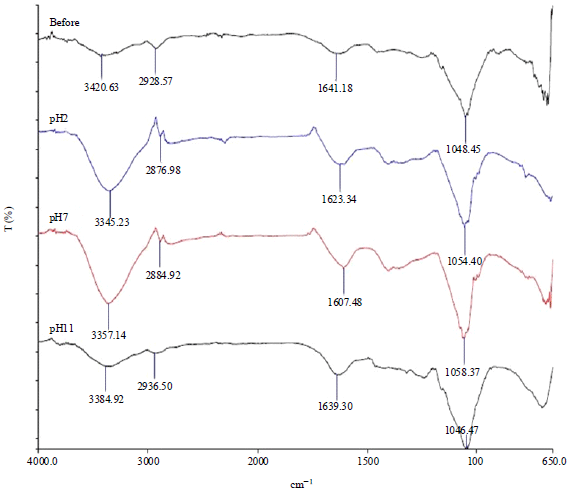
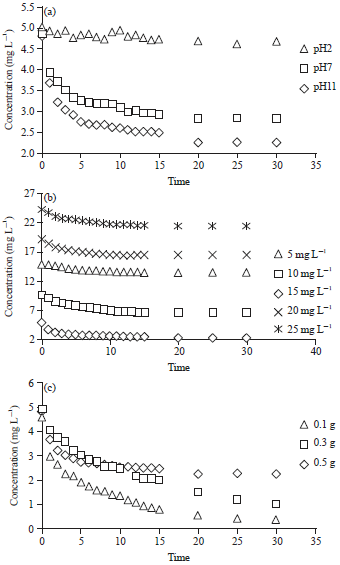
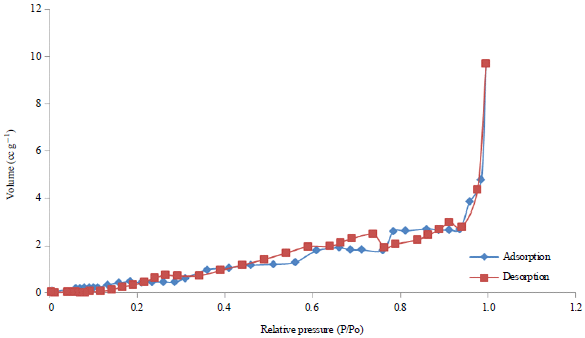
Research Article
Parametric and Adsorption Kinetic Studies of Reactive Black 5 Removal from Textile Simulated Wastewater Using Oil Palm (Elais guineensis) Empty Fruit Bunch
Sustainable Palm Oil Research Unit, Faculty of Science and Natural Resources, Universiti Malaysia Sabah, Kota Kinabalu, Sabah, 88400, Malaysia
Wan Mohd Ashri Wan Daud
Department of Chemical Engineering, Faculty of Engineering, University of Malaya, Kuala Lumpur, 50603, Malaysia
Quek Kye Shane
Sustainable Palm Oil Research Unit, Faculty of Science and Natural Resources, Universiti Malaysia Sabah, Kota Kinabalu, Sabah, 88400, Malaysia
Kogularama Sanmugam
Sustainable Palm Oil Research Unit, Faculty of Science and Natural Resources, Universiti Malaysia Sabah, Kota Kinabalu, Sabah, 88400, Malaysia